| Citation: | Xi Yan, Junkui Shang, Runrun Wang, Fengyu Wang, Jiewen Zhang. Mechanisms regulating cerebral hypoperfusion in cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy[J]. The Journal of Biomedical Research, 2022, 36(5): 353-357. DOI: 10.7555/JBR.36.20220208 |
Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) is a common pure form of subcortical vascular dementia, which is caused by NOTCH3 gene mutation. Granular osmiophilic material (GOM) deposition in the small arteries is the classical pathology of CADASIL[1]. White matter hyperintensity (WMH), especially that in the anterior temporal lobes and external capsule, has been identified as a typical neuroimaging feature of CADASIL[2]. Currently, many mechanisms are reported to be linked to WMH, including cerebral hypoperfusion, impaired cerebral vascular reactivity, and blood-brain barrier impairment. Among them, cerebral hypoperfusion is an early feature of the disease and its relationship with WMH has been clearly investigated by many groups[3]. Collectively, these studies provide strong evidence demonstrating that cerebral hypoperfusion is an early event and may play a key role in the CADASIL pathophysiological cascade[4]. In this review, we summarize the possible pathophysiology of hypoperfusion in CADASIL.
Although there is sufficient evidence confirmed the existence of cerebral hemodynamic abnormalities and cerebral arteriolar dysfunction in CADASIL patients, how NOTCH3 gene mutation causes arteriolar dysfunction has not been clearly studied. The mature Notch3 receptor consists of the 210-kDa Notch3 extracellular domain (Notch3ECD) and the 97-kDa Notch3 transmembrane intracellular domain (Notch3TICD). Signal activation of Notch3 requires the Notch3 receptor to undergo a series of proteolytic cleavages for division into Notch3ECD and Notch3TICD[5]. Notch3 protein is mainly expressed in mural cells, including vascular smooth muscle cells (VSMCs) and pericytes[6]. Therefore, it can be speculated that abnormal vascular mural cells may be an important reason for the dysfunction of arterioles in CADASIL.
Accumulation of Notch3ECD is one of the earliest detectable pathological features in CADASIL patients and mouse models[4], suggesting that Notch3ECD accumulation is an early event in CADASIL. NOTCH3 mutations lead to an odd number of cysteine residues in Notch3ECD, consequently resulting in abnormal degradation of Notch3ECD on the surface of mural cells. Joutel A. is the first to put forward the Notch3ECD cascade hypothesis for CADASIL: early Notch3ECD deposition abnormally recruits the other extracellular matrix (ECM) proteins, thus leading to the accumulation and formation of the so-called GOM, which can be observed under the transmission electron microscope[7]. Notch3ECD along with other ECM proteins causes dysfunction in the small vessels of the brain. This hypothesis provides an alternate understanding of the pathogenesis of CADASIL. Postmortem studies have found a series of ECM proteins that are components of GOM, including clusterin, tissue inhibitor of metalloproteinases 3 (TIMP3), vitronectin, latency in TGF-β binding protein 1 (LTBP-1), high-temperature requirement protein A1 (HTRA1), and other ECM proteins[8]. Abnormal deposition of these proteins can affect the normal structure and function of arterioles. TIMP3 is a tissue inhibitor of matrix metalloproteinase, which helps maintain tissue microenvironment homeostasis[9]. Studies have shown that the increase of TIMP3 is associated with abnormal small artery function and decreased cerebral blood flow (CBF) in mouse models[10]. Abnormal TIMP3 accumulation inactivates the metalloprotease ADAM17, which results in the inactivation of HB-EGF-ErbB1/ErbB4 signaling and endocytic dysfunction of the voltage-dependent potassium channels (Kv). Increased Kv channel activity in the VSMCs membrane leads to compromised cerebral arterial tone and CBF responses, which can be restored by exogenous addition of ADAM17 or HB-EGF[11]. Besides, an autophagy-lysosomal defect was detected in human CADASIL VSMCsR133C[12], aggravating the accumulation of intracellular and extracellular proteins. Abnormal aggregation and deposition of proteins can further lead to endoplasmic reticulum oxidative stress, apoptosis and cell dysfunction[13].
Notch3 signaling plays an important role in arterial maturation, maintenance, and function[14]. Given its critical role in vascular integrity, dysregulation of Notch3 signaling may be associated with cerebrovascular dysfunction in individuals with NOTCH3 gene mutations. Severe cerebrovascular abnormalities have been detected in NOTCH3 gene knockout mice, including VSMC loss, blood-brain barrier leakage, and arterial myogenic tone changes[14]. Interestingly, cerebrovascular abnormalities reported in NOTCH3 knockout mice were consistent with those observed in CADASIL mutant mice (TgNotch3C455R)[15]. However, except for mutations in the binding region of Notch3 receptor, the Notch3 pathway activity is not significantly affected[16–17]. Besides, increased Notch3 activity and the impairment of maximal vasodilator capacity were found in CADASIL mutant mice (TgNotch3R169C), which can be prevented by conditionally reducing Notch3 activity and mimicked by conditionally activating Notch3 in VSMCs[18]. Therefore, different NOTCH3 mutations may have different effects on Notch3 signaling, and contribute to reductions in CBF through different mechanisms.
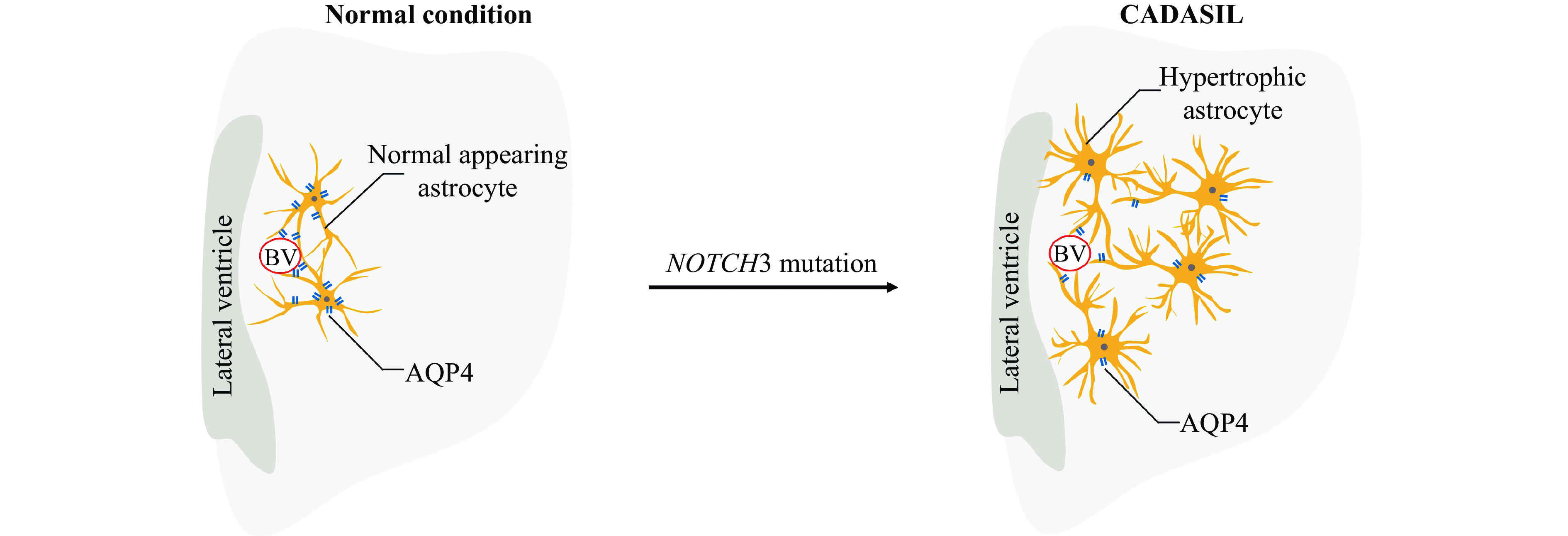
In addition to mural cells, Notch3 receptor is also expressed in astrocytes. Being a part of the cellular composition of the neurovascular unit (NVU), astrocytes contribute to the regulation of small vessel tone and CBF responses[19]. In 2010, Brennan-Krohn T et al reported that astrocytic-associated molecular abnormalities are restricted to the regions of WMH and contribute to vascular degeneration in brain tissues in patients with CADASIL[20]. Furthermore, in 2018, Hase Y et al demonstrated the activation of normal-appearing astrocytes and their transformation into clasmatodendritic astrocytes in the deep white matter in CADASIL[21] (Fig. 1). Immunohistochemical analyses have shown that clasmatodendritic astrocytes are abundant in the anterior temporal pole and that aquaporin 4 (AQP4) is displaced in these astrocytes[21], indicating that the gliovascular unit of the deep white matter is severely impaired in CADASIL (Fig. 1). Next, we tried to understand the pathology of neurovascular dysfunction and WMH in light of astrocytopathy in CADASIL.
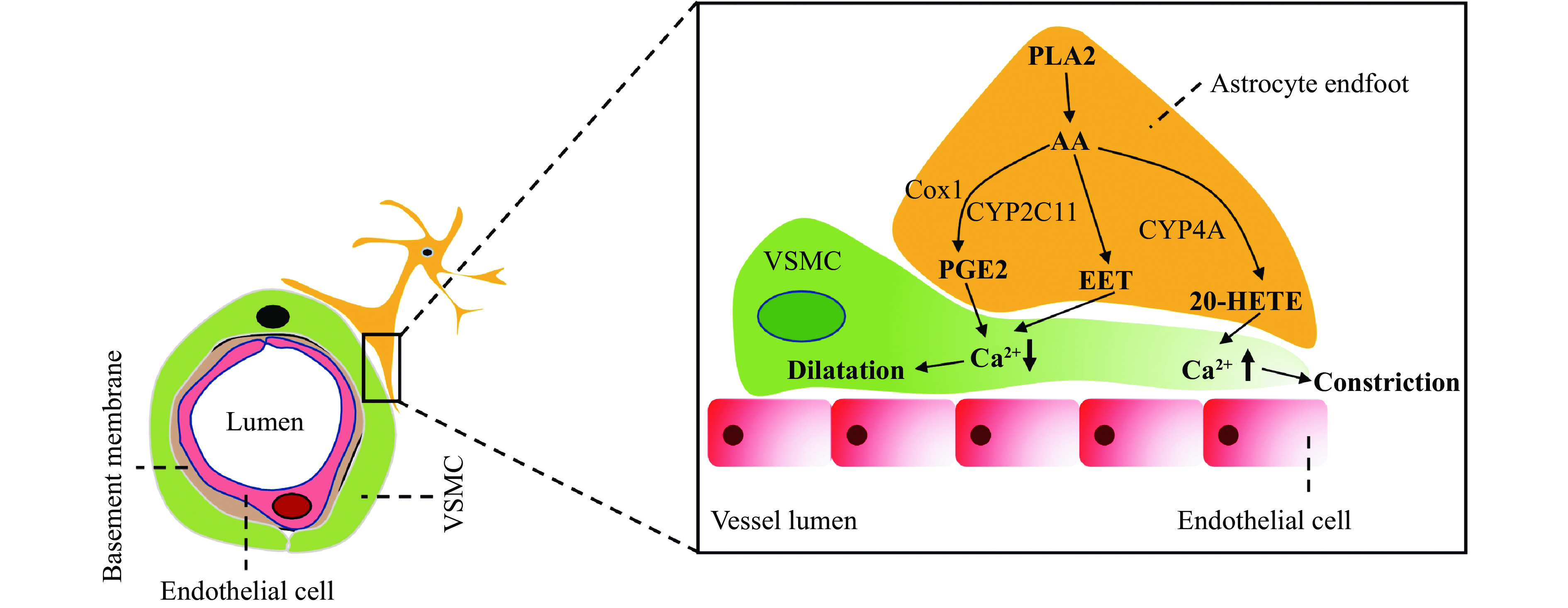
Astrocyte endfeet show close apposition around VSMCs and pericytes. Increased intracellular Ca2+ levels in astrocyte endfeet result in arteriolar vasodilation[22]. A study has reported the astrocyte-regulated vascular tone in VSMC through the phospholipase A2-arachidonic acid pathway[23] (Fig. 2). Whether astrocytopathy-mediated VSMC dysfunction is the cause of CBF reduction in CADASIL remains unknown.
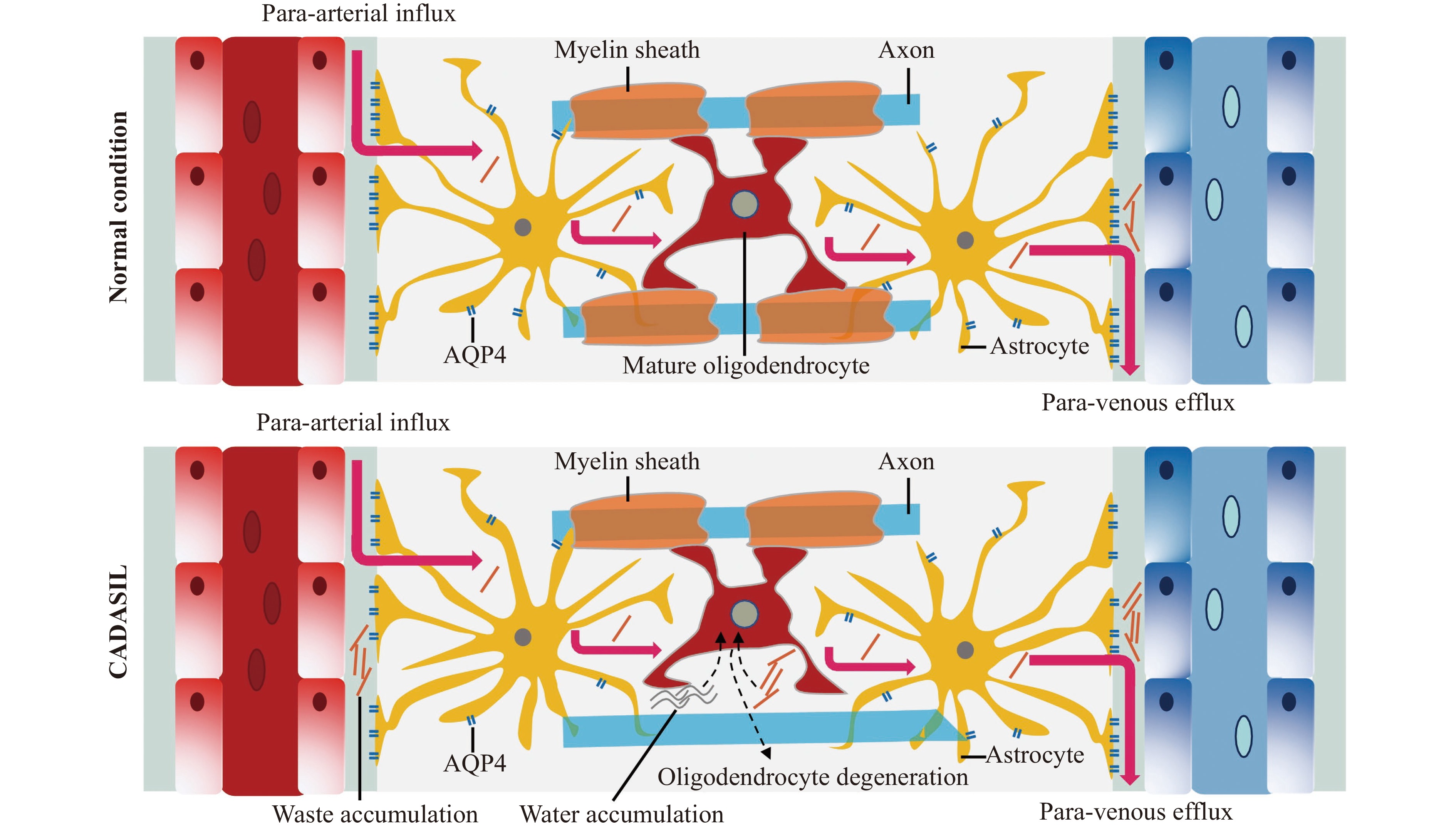
Astrocyte endfeet also form a unique network of interconnected perivascular tunnels called the glymphatic system[24] (Fig. 3). The function of the perivascular glymphatic pathway is primarily dependent on AQP4 water channels, which are highly expressed in the astrocyte endfeet. The glymphatic system not only facilitates the elimination of excess fluid and interstitial waste in the brain parenchyma but also plays an important role in the fluid exchange between cerebrospinal fluid (CSF) and brain parenchyma interstitial fluid (ISF). Waste clearance and fluid exchange are critical for brain homeostasis maintenance[24]. WMH represents local fluid accumulation in the white matter. Decreased glymphatic activity probably results in waste and fluid accumulation and further initiates the loss of mature oligodendrocytes and WMH in patients with CADASIL[25] (Fig. 3). WMH is not only the hallmark of CADASIL, but also the characteristic pathological change in other small-vessel diseases (SVDs). The stroke-prone spontaneously hypertensive rat (SHRSP) is a rodent model of human sporadic SVD. Studies show that total CSF flux is significantly reduced in the SHRSP rats than in controls. The glymphatic flux is also significantly reduced in SHRSP rats than in controls. Altered perivascular AQP4 expression is also observed in SHRSP[26]. Diabetes is a cardiovascular risk factor for SVD. Dysfunctional CSF flow dynamics and glymphatic transport occur in the streptozotocin-induced type 2 diabetes rat model[27].
Recent studies have demonstrated that CBF hemodynamic deficit occurs in the early stage of CADASIL and plays an important role in brain parenchymal injury formation. As the component of NVU, endothelial cell, VSMC, pericyte, astrocyte and ECM all participate in CBF regulation. We reviewed the possible pathophysiology of hypoperfusion in CADASIL from the aspects of mural cells and glial cells. NOTCH3 gene mutations cause abnormal Notch3 deposition and signaling, leading to mural cell dysfunction and degeneration. However, whether severe astrocytopathy is the main cause of vascular degeneration or it is secondary to VSMC- and pericyte-mediated vascular pathology in the deep white matter of CADASIL remains to be elucidated. Much more work is needed to elucidate the relationship between NOTCH3 mutations and decreased CBF in order to seek therapeutic targets and clinical translations.
This work was supported by National Natural Science Foundation of China (Grants No. 81873727 and 82171196).
CLC number: R743, Document code: A
The authors reported no conflict of interests.
| [1] |
Di Donato I, Bianchi S, De Stefano N, et al. Cerebral Autosomal Dominant Arteriopathy with Subcortical Infarcts and Leukoencephalopathy (CADASIL) as a model of small vessel disease: update on clinical, diagnostic, and management aspects[J]. BMC Med, 2017, 15(1): 41. doi: 10.1186/s12916-017-0778-8
|
| [2] |
Schoemaker D, Quiroz YT, Torrico-Teave H, et al. Clinical and research applications of magnetic resonance imaging in the study of CADASIL[J]. Neurosci Lett, 2019, 698: 173–179. doi: 10.1016/j.neulet.2019.01.014
|
| [3] |
Huneau C, Houot M, Joutel A, et al. Altered dynamics of neurovascular coupling in CADASIL[J]. Ann Clin Transl Neurol, 2018, 5(7): 788–802. doi: 10.1002/acn3.574
|
| [4] |
Joutel A, Monet-Leprêtre M, Gosele C, et al. Cerebrovascular dysfunction and microcirculation rarefaction precede white matter lesions in a mouse genetic model of cerebral ischemic small vessel disease[J]. J Clin Invest, 2010, 120(2): 433–445. doi: 10.1172/JCI39733
|
| [5] |
Mašek J, Andersson ER. The developmental biology of genetic Notch disorders[J]. Development, 2017, 144(10): 1743–1763. doi: 10.1242/dev.148007
|
| [6] |
Liu H, Zhang W, Kennard S, et al. Notch3 is critical for proper angiogenesis and mural cell investment[J]. Circ Res, 2010, 107(7): 860–870. doi: 10.1161/CIRCRESAHA.110.218271
|
| [7] |
Monet-Leprêtre M, Haddad I, Baron-Menguy C, et al. Abnormal recruitment of extracellular matrix proteins by excess Notch3ECD: a new pathomechanism in CADASIL[J]. Brain, 2013, 136(6): 1830–1845. doi: 10.1093/brain/awt092
|
| [8] |
Zellner A, Scharrer E, Arzberger T, et al. CADASIL brain vessels show a HTRA1 loss-of-function profile[J]. Acta Neuropathol, 2018, 136(1): 111–125. doi: 10.1007/s00401-018-1853-8
|
| [9] |
Fan D, Kassiri Z. Biology of tissue inhibitor of metalloproteinase 3 (TIMP3), and its therapeutic implications in cardiovascular pathology[J]. Front Physiol, 2020, 11: 661. doi: 10.3389/fphys.2020.00661
|
| [10] |
Capone C, Cognat E, Ghezali L, et al. Reducing Timp3 or vitronectin ameliorates disease manifestations in CADASIL mice[J]. Ann Neurol, 2016, 79(3): 387–403. doi: 10.1002/ana.24573
|
| [11] |
Capone C, Dabertrand F, Baron-Menguy C, et al. Mechanistic insights into a TIMP3-sensitive pathway constitutively engaged in the regulation of cerebral hemodynamics[J]. Elife, 2016, 5: e17536. doi: 10.7554/eLife.17536
|
| [12] |
Hanemaaijer ES, Panahi M, Swaddiwudhipong N, et al. Autophagy-lysosomal defect in human CADASIL vascular smooth muscle cells[J]. Eur J Cell Biol, 2018, 97(8): 557–567. doi: 10.1016/j.ejcb.2018.10.001
|
| [13] |
Neves KB, Morris HE, Alves-Lopes R, et al. Peripheral arteriopathy caused by Notch3 gain-of-function mutation involves ER and oxidative stress and blunting of NO/sGC/cGMP pathway[J]. Clin Sci (Lond), 2021, 135(6): 753–773. doi: 10.1042/CS20201412
|
| [14] |
Henshall TL, Keller A, He L, et al. Notch3 is necessary for blood vessel integrity in the central nervous system[J]. Arterioscler Thromb Vasc Biol, 2015, 35(2): 409–420. doi: 10.1161/ATVBAHA.114.304849
|
| [15] |
Machuca-Parra AI, Bigger-Allen AA, Sanchez AV, et al. Therapeutic antibody targeting of Notch3 signaling prevents mural cell loss in CADASIL[J]. J Exp Med, 2017, 214(8): 2271–2282. doi: 10.1084/jem.20161715
|
| [16] |
Monet-Leprêtre M, Bardot B, Lemaire B, et al. Distinct phenotypic and functional features of CADASIL mutations in the Notch3 ligand binding domain[J]. Brain, 2009, 132(6): 1601–1612. doi: 10.1093/brain/awp049
|
| [17] |
Monet M, Domenga V, Lemaire B, et al. The archetypal R90C CADASIL-NOTCH3 mutation retains NOTCH3 function in vivo[J]. Hum Mol Genet, 2007, 16(8): 982–992. doi: 10.1093/hmg/ddm042
|
| [18] |
Baron-Menguy C, Domenga-Denier V, Ghezali L, et al. Increased Notch3 activity mediates pathological changes in structure of cerebral arteries[J]. Hypertension, 2017, 69(1): 60–70. doi: 10.1161/HYPERTENSIONAHA.116.08015
|
| [19] |
Kisler K, Nelson AR, Montagne A, et al. Cerebral blood flow regulation and neurovascular dysfunction in Alzheimer disease[J]. Nat Rev Neurosci, 2017, 18(7): 419–434. doi: 10.1038/nrn.2017.48
|
| [20] |
Brennan-Krohn T, Salloway S, Correia S, et al. Glial vascular degeneration in CADASIL[J]. J Alzheimers Dis, 2010, 21(4): 1393–1402. doi: 10.3233/JAD-2010-100036
|
| [21] |
Hase Y, Chen A, Bates LL, et al. Severe white matter astrocytopathy in CADASIL[J]. Brain Pathol, 2018, 28(6): 832–843. doi: 10.1111/bpa.12621
|
| [22] |
Mulligan SJ, MacVicar BA. Calcium transients in astrocyte endfeet cause cerebrovascular constrictions[J]. Nature, 2004, 431(7005): 195–199. doi: 10.1038/nature02827
|
| [23] |
Takano T, Tian G, Peng W, et al. Astrocyte-mediated control of cerebral blood flow[J]. Nat Neurosci, 2006, 9(2): 260–267. doi: 10.1038/nn1623
|
| [24] |
Jessen NA, Munk AS, Lundgaard I, et al. The glymphatic system: a beginner's guide[J]. Neurochem Res, 2015, 40(12): 2583–2599. doi: 10.1007/s11064-015-1581-6
|
| [25] |
Benveniste H, Nedergaard M. Cerebral small vessel disease: a glymphopathy?[J]. Curr Opin Neurobiol, 2022, 72: 15–21. doi: 10.1016/j.conb.2021.07.006
|
| [26] |
Koundal S, Elkin R, Nadeem S, et al. Optimal mass transport with lagrangian workflow reveals advective and diffusion driven solute transport in the glymphatic system[J]. Sci Rep, 2020, 10(1): 1990. doi: 10.1038/s41598-020-59045-9
|
| [27] |
Jiang Q, Zhang L, Ding G, et al. Impairment of the glymphatic system after diabetes[J]. J Cereb Blood Flow Metab, 2017, 37(4): 1326–1337. doi: 10.1177/0271678X16654702
|
| [1] | Runrun Wang, Jiewen Zhang, Junkui Shang, Fengyu Wang, Xi Yan. Effects of different regional cerebral blood flow on white matter hyperintensity in CADASIL patients[J]. The Journal of Biomedical Research, 2022, 36(5): 368-374. DOI: 10.7555/JBR.36.20220006 |
| [2] | Qian Sun, Yusi Hu, Saiyue Deng, Yanyu Xiong, Zhili Huang. A visualization pipeline for in vivo two-photon volumetric astrocytic calcium imaging[J]. The Journal of Biomedical Research, 2022, 36(5): 358-367. DOI: 10.7555/JBR.36.20220099 |
| [3] | Xiaolong Zheng, Wei Wang. Astrocyte transplantation for repairing the injured spinal cord[J]. The Journal of Biomedical Research, 2022, 36(5): 312-320. DOI: 10.7555/JBR.36.20220012 |
| [4] | Marta Obara-Michlewska. The contribution of astrocytes to obesity-associated metabolic disturbances[J]. The Journal of Biomedical Research, 2022, 36(5): 299-311. DOI: 10.7555/JBR.36.20200020 |
| [5] | Tiwari-Heckler Shilpa, Jiang Z. Gordon, Popov Yury, J. Mukamal Kenneth. Daily high-dose aspirin does not lower APRI in the Aspirin-Myocardial Infarction Study[J]. The Journal of Biomedical Research, 2020, 34(2): 139-142. DOI: 10.7555/JBR.33.20190041 |
| [6] | Huan Liu, Shijiang Zhang, Yongfeng Shao, Xiaohu Lu, Weidong Gu, Buqing Ni, Qun Gu, Junjie Du. Biomechanical characterization of a novel ring connector for sutureless aortic anastomosis[J]. The Journal of Biomedical Research, 2018, 32(6): 454-460. DOI: 10.7555/JBR.31.20170011 |
| [7] | Minbo Zang, Qiao Zhou, Yunfei Zhu, Mingxi Liu, Zuomin Zhou. Effects of chemotherapeutic agent bendamustine for nonhodgkin lymphoma on spermatogenesis in mice[J]. The Journal of Biomedical Research, 2018, 32(6): 442-453. DOI: 10.7555/JBR.31.20170023 |
| [8] | Huanqiang Wang, Congying Yang, Siyuan Wang, Tian Wang, Jingling Han, Kai Wei, Fucun Liu, Jida Xu, Xianzhen Peng, Jianming Wang. Cell-free plasma hypermethylated CASZ1, CDH13 and ING2 are promising biomarkers of esophageal cancer[J]. The Journal of Biomedical Research, 2018, 32(6): 424-433. DOI: 10.7555/JBR.32.20170065 |
| [9] | Fengzhen Wang, Mingwan Zhang, Dongsheng Zhang, Yuan Huang, Li Chen, Sunmin Jiang, Kun Shi, Rui Li. Preparation, optimization, and characterization of chitosancoated solid lipid nanoparticles for ocular drug delivery[J]. The Journal of Biomedical Research, 2018, 32(6): 411-423. DOI: 10.7555/JBR.32.20160170 |
| [10] | Sundeep?S.?Tumber, Hong?Liu. Epidural abscess after multiple lumbar punctures for labour epidural catheter placement[J]. The Journal of Biomedical Research, 2010, 24(4): 332-335. DOI: 10.1016/S1674-8301(10)60046-2 |
| 1. | Sánchez-Lanzas R, Barclay J, Hardas A, et al. A CADASIL NOTCH3 mutation leads to clonal hematopoiesis and expansion of Dnmt3a-R878H hematopoietic clones. Leukemia, 2024. DOI:10.1038/s41375-024-02464-8. Online ahead of print |
| 2. | Anisetti B, Greco E, Stojadinovic E, et al. Novel grading system for CADASIL severity: A multicenter cross-sectional study. Cereb Circ Cogn Behav, 2023, 5: 100170. DOI:10.1016/j.cccb.2023.100170 |
| 3. | Muppa J, Yaghi S, Goldstein ED. Antiplatelet use and CADASIL: a retrospective observational analysis. Neurol Sci, 2023, 44(8): 2831-2834. DOI:10.1007/s10072-023-06773-1 |
| 4. | Ding F. Editorial commentary on the special issue of glia and neurological diseases. J Biomed Res, 2022, 36(5): 297-298. DOI:10.7555/JBR.36.20220800 |