| Citation: | Evgenii Dmitrievich Kryl'skii, Darya Andreevna Sinitsyna, Tatyana Nikolaevna Popova, Khidmet Safarovich Shikhaliev, Svetlana Mikhajlovna Medvedeva, Larisa Vladimirovna Matasova, Valentina Olegovna Mittova. The new antioxidant 1-benzoyl-6-hydroxy-2,2,4-trimethyl-1,2-dihydroquinoline has a protective effect against carbon tetrachloride-induced hepatic injury in rats[J]. The Journal of Biomedical Research, 2022, 36(6): 423-434. DOI: 10.7555/JBR.36.20220098 |
Liver disease remains one of the ten leading causes of death worldwide[1]. When the liver is exposed to toxins, including carbon tetrachloride (CCl4), metabolic dysfunction can occur, causing liver fibrosis, cirrhosis, or hepatocellular carcinoma in some instances[2]. CCl4 injections are thought to generate reactive molecules with cytochrome P450 2E1 (Cyp2E1) and through trichloromethyl peroxyl radical formation[3]. The resulting oxidative stress can promote lipid peroxidation and the damage of hepatocellular membrane, release of proinflammatory chemokines and cytokines and development of inflammation and apoptosis[4]. Therefore, protecting the liver from oxidative stress is necessary and can be initiated with antioxidant enzymes superoxide dismutase (SOD) and catalase. Likewise, glutathione peroxidase (GP) can oxidize glutathione (GSH) which reduces levels of organic and inorganic peroxides. The reduction of oxidized glutathione (GSSG) can also be catalysed by glutathione reductase (GR). This means that glutathione S-transferases (GSTs) found in liver cells, can be considered the main group of protective enzymes against chemical stress caused by toxins and carcinogenesis[5].
Despite the large number of hepatoprotective agents, they all have some disadvantages. For example, silymarin loses its importance in acute liver damage due to low bioavailability and the need for long-term use by patients[6]. Unfortunately, silybin is not particularly water soluble, and is not easily absorbed through the intestines[7]. Similarly, ursodeoxycholic acid which can be used as an intervention has also been associated with difficulties similar to those encountered with silybin[7]. An alternative is sulpho-adenosylmethionine which has been trialled and appears to cause anxiety and diarrhoea in some patients[8]. So, despite having relatively manageable side effects these interventions are not considered appropriate. Vitamin E is another potential alternative although there are no compositional standards and the pro-oxidant effects caused by high concentrations are concerns[9]. Finally, selonsertib has been shown to reduce the degree of bridging fibrosis and cirrhosis, but is again associated with significant adverse events[9]. Therefore, we know which mechanisms target to improve outcomes but we do not yet have a safe and effective intervention.
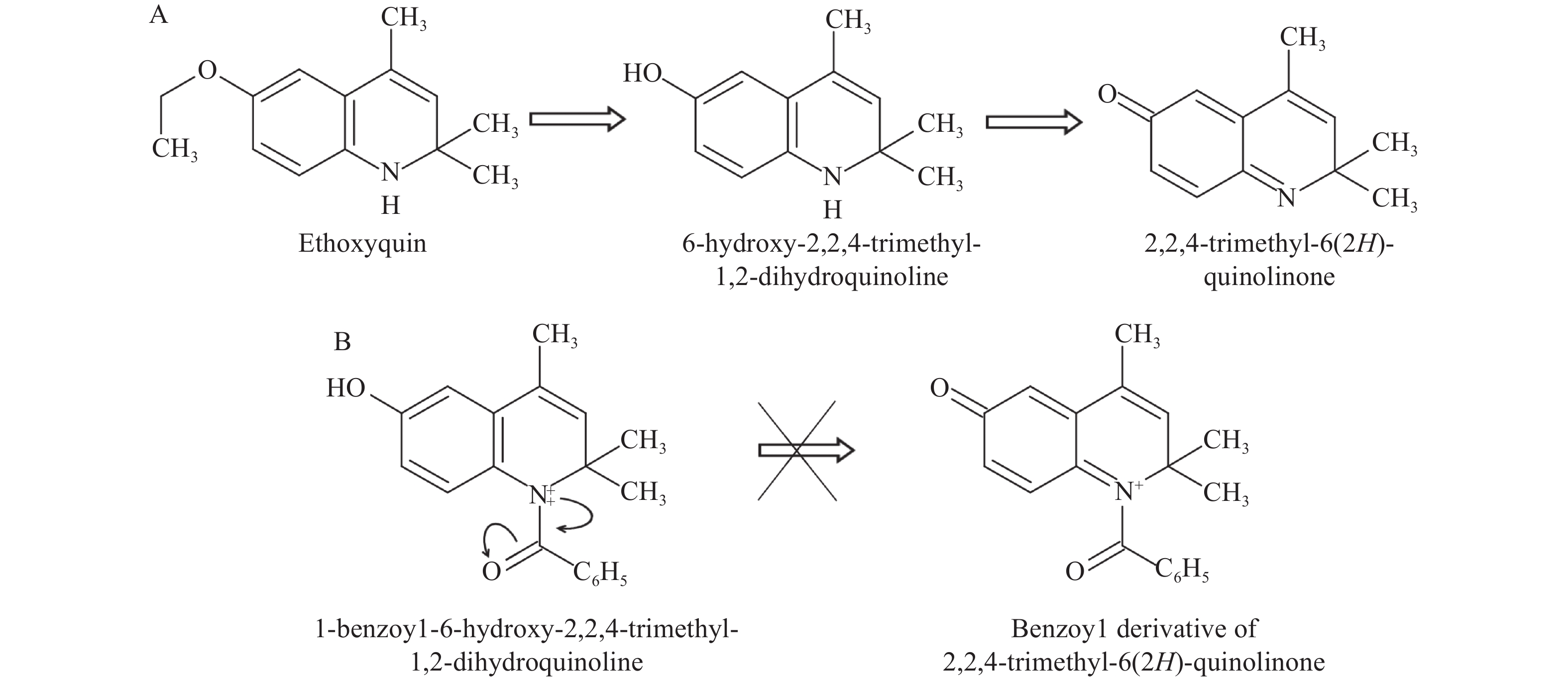
Given the central role of oxidative stress in the pathogenesis of toxic liver damage, research into the hepatoprotective effect of substances with high antioxidant potential would seem necessary. Compounds including synthetic antioxidant ethoxyquin have not been approved for use in humans. In a previous study, we demonstrated the hepatoprotectivity of a derivative, 6-hydroxy-2,2,4-trimethyl-1,2-dihydroquinoline[10]. After conducting that study, we inserted a benzoyl group into the structure of this compound and created a new derivative, 1-benzoyl-6-hydroxy-2,2,4-trimethyl-1,2-dihydroquinoline (BDHQ). BHDQ has the potential to prevent the formation of a toxic 2,2,4-trimethyl-6(2H)-quinolinone from the compound (Fig. 1)[11].
Therefore, we assessed the antioxidization and hepatoprotective properties of BHDQ in rodents with acute liver damage. Here, we tested a dihydroquinoline derivative in terms of hepatocyte cytolysis markers and histopathologic changes in the liver. We also looked to describe how BHDQ inhibits the development of oxidative stress and modulates antioxidation in rats with CCl4-induced liver damage. Finally, we compared silymarin to BHDQ in terms of hepatoprotectivity and antioxidization.
BHDQ was synthesized according to a previously developed scheme[11]. The LD50 of the compound was 800 mg/kg body weight (BW). Diagnostic kits B 01.01 ALT-VITAL, B 02.01 AST-VITAL and B 17.11 Triglycerides-Vital were obtained from Vital Development Corporation (Russia). Diagnostic kit 007.004 Gamma-GT-Olvex was purchased from Olvex Diagnosticum (Russia). 1-chloro-2,4-dinitrobenzene acid and 5,5′-dithiobis (2-nitrobenzoic acid) were obtained from Sigma Aldrich (USA). Citrate was purchased from PanReac (Spain). GSH, NADPH and nitroblue tetrazolium (NBT) were from AppliChem (Germany). All products related to polymerase chain reaction (PCR) were from Eurogen (Russia). Caspase-8 and caspase-3 kits were obtained from Abcam (UK). BCA Protein Assay Kit Ⅱ was obtained from BioVision (USA).
Sixty male Wistar rats, 4–6 months old and weighing 200–250 g, were housed under normal light-dark conditions (12 hours light followed by 12 hours dark) for the entire experiment and had access to food and water, ad libitum. The food comprises the ingredients of: barley, oats, wheat bran, meat and bone meal, table salt, and lime flour. The nutrient content of the laboratory animal diets was shown in Supplementary Table 1 (available online).
The study protocol was approved by the Ethics committee on biomedical research expertise in Voronezh State University (Voronezh, Russia) and was conducted in accordance with the EU directive 2010/63/EU for animal experiments.
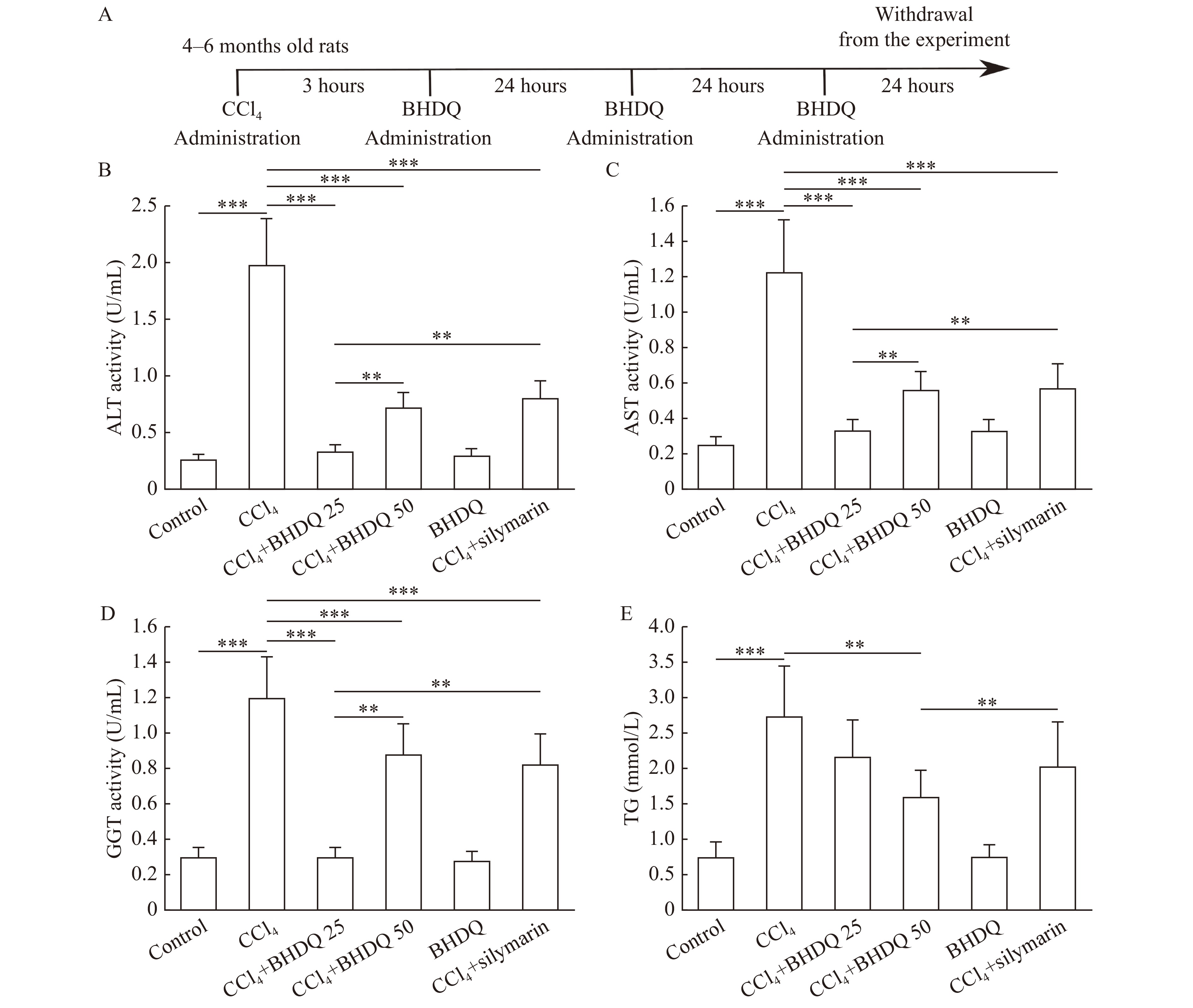
The animals were randomly assigned to either a control, CCl4, CCl4+BHDQ 25, CCl4+BHDQ 50, BHDQ, or a CCl4+silymarin group. Each group consisted of 10 rodent subjects. The control group received a single dose of 1 mL of vaseline oil via oral gavage. Rats in the CCl4 group received a single intragastric dose of CCl4 (64 μL dissolved in 1 mL of vaseline oil per 100 g BW)[10]. Rats in CCl4+BHDQ 25 and CCl4+BHDQ 50 groups received BHDQ (25 and 50 mg respectively, dissolved in 1 mL of 1% soluble starch solution per 1 kg BW) 3 hours after the administration of CCl4 and was administrated every 24 hours for three days (Fig. 2A). Rats in the CCl4+silymarin group were treated with 50 mg/kg BW silymarin (Carsil, Sopharma, Bulgaria) after the administration of CCl4 under the same scheme. Rats in the BHDQ group were treated with BHDQ (50 mg/kg BW) once per day for three days. The dosage of BHDQ was calculated according to the dosage of Carsil given in the instructions for use. In accordance with the instructions, Carsil is administered for 90 days at a dose of 105–210 mg per day, corresponding to 135 to 270 mg/kg of the drug per 70-kg person. Accordingly, in the CCl4+BHDQ 50 and BHDQ groups, animals received a total of 150 mg/kg BW BHDQ over the entire experimental period.
All animals were humanely euthanized on day 4 to harvest blood and liver tissues. On the fourth day from the beginning of the experiment, rats were sacrificed, liver and blood were extracted and immediately used for biochemical analysis.
Liver homogenate and blood serum were used in further studies. Parts of the liver of each rat were frozen at −80 °C for PCR analysis. Each investigated indicator in the samples was analyzed in triplicate.
Hematoxylin and eosin (H&E) staining of liver was assessed for three rats from each group. Rats were anesthetized with 5 mL chloroform in a soaked cotton bud, in the vacuum sealed glass chamber for 2 minutes[12]. Livers were rapidly removed and immersed in 10% formalin for 2 hours, then washed three times using PBS. After being dehydrated and embedded with paraffin, liver tissues were sliced into 6-µm-thick coronal sections using a rotary microtome HM-325 (Thermo Fisher Scientific, USA) for the H&E staining. High magnification images were captured using an AxioLab A1 light microscope (Zeiss, Germany). A minimum of five fields for each slide were assessed. A numerical scoring for assessing histological activity was carried out in accordance with the approach reported by Knodell et al[13].
Oxidative stress intensity and the total antioxidant activity in liver and blood serum were measured through biochemiluminescence (BChL), induced by hydrogen peroxide with iron sulfate[14]. This method is based on the decomposition of H2O2 by transition valence metal ions (Fe2+) in the Fenton reaction. Generating reactive oxygen species (ROS) initiates oxidation in samples. Recombining formed radicals generate unstable tetroxide, which in turn decomposes with the emission of a quantum of light.
The reaction medium contained 0.4 mL of 0.02 mmol/L potassium phosphate buffer (pH 7.5), 0.4 mL of 0.01 mmol/L FeSO4 and 0.2 mmol/L of a 2% H2O2 solution introduced immediately before measurement. The test sample was added in 0.1 mL amounts before adding the 2% H2O2 solution. The BChL kinetic curve was recorded for 30 seconds using a BChL-07 biochemiluminometer (Medozons, Russia). The following parameters were determined: the light sum of chemiluminescence (S) and maximum intensity (Imax) was used to characterize the level of oxidative stress, and tangent of the BChL kinetic curve slope (tgα2) was used to characterize characterizing antioxidant activity.
The activities of alanine aminotransferase (ALT), aspartate aminotransferase (AST), gamma-glutamyl transpeptidase (GGT) and concentration of triglycerides (TG) were measured using diagnostic kits listed in reagents. Concentrations of diene conjugates (DC), citrate and GSH were analyzed as described earlier[11].
Aconitate hydratase (AH) activity was determined spectrophotometrically at 235 nm. The medium for the AH activity assay included: 50 mmol/L Tris-HCl buffer, pH 7.8, 4 mmol/L citrate[15]. Reactions were initiated by introducing the test sample to a spectrophotometric medium.
SOD activity was measured at 540 nm based on NBT reduction[16]. Catalase activity was assessed according to the method developed by Góth[17]. GP activity was assayed according to Paglia and Valentina[18]. GR activity was assessed by measuring the oxidation of NADPH (0.16 mmol/L) using oxidized glutathione as a substrate[19]. GT activity was assessed using GSH and 1-chloro-2,4-dinitrobenzene acid as substrates[20]. Glucose-6-phosphate dehydrogenase (G6PD) and NADP-isocitrate dehydrogenase (NADP-IDH) activity was measured as described previously[11].
Enzyme activities of caspase-8 and caspase-3 were measured using colorimetric assay kits according to manufacturer's protocols. The enzymatic activity was evaluated using a Hitachi U1900 spectrophotometer (Japan).
Total RNA was isolated from liver tissue using ExtractRNA reagent (Eurogen, Russia). Quantitative reverse transcription PCR was performed using qPCRmix-HS SYBR with a BioRad Connect device (BioRad, USA) according to the manufacture's instruction. The mRNA level of each gene was normalized to house-keeping genes such as Gapdh and Actb. Primers were listed in Supplementary Table 2 (available online).
Multiple groups were analyzed using a one-way ANOVA with Tukey's post hoc test, P<0.05 was considered statistically significant. Statistical analysis was performed using the IBM SPSS Statistics (version 25.0) software. All quantitative data are presented as mean±SD.
We found that CCl4-induced hepatic injury was associated with an increase in ALT (P<0.001), AST (P<0.001), and GGT (P<0.001) activity in blood serum of animals compared to the control group. There was also an increase in TG concentration in the liver of animals with CCl4-induced hepatic injury compared to the control group (P<0.001). At the same time, the administration of BHDQ with CCl4-induced hepatic injury decreased the level of ALT (P<0.001), AST (P<0.001), and GGT (P<0.001) compared to the CCl4 group, and the dose of BHDQ of 25 mg/kg BW appeared more effective (P=0.002 for ALT and AST, P=0.001 for GGT) (Fig. 2B–E).
Silymarin did not differ from that of BHDQ in terms of efficacy, in relation to liver functions, at a dose of 50 mg/kg BW. However, the effectiveness of silymarin was lower than BHDQ, at a dose of 25 mg/kg BW (P=0.003 for ALT and AST, P=0.001 for GGT). There was also a significant reduction (P=0.001) in TG concentrations which appears to have been facilitated by administering the BHDQ at a dose of 50 mg/kg BW, compared to the CCl4 group (Fig. 2E).
Histological analysis of liver tissue morphologies confirmed the protective effect of BHDQ in CCl4-induced hepatic injury. The liver in the control group showed normal hepatocytes, obvious sinusoids (S), and central vein (CV) (Fig. 3A and B). Liver tissues in the CCl4 group was characterized by focal necrotic cell death (N), diffuse fatty changes (FC), microvesicular steatosis in hepatocyte cytosol (MS), and inflammatory infiltration (II) (Fig. 3C and D). Livers in the CCl4+BDHQ 25 (Fig. 3E and F) and CCl4+silymarin (Fig. 3K and L) groups showed less severe liver injury with some pathological fatty deposition (FD) and focal hepatocellular degeneration (FHD). No fatty liver dysplasia was observed in the CCl4+BDHQ 50 (Fig. 3G and H) group. BDHQ group indicated no pathologic lesions (Fig. 3I and J). A numerical scoring for histopathological changes is present in Fig. 3M. Therefore, BHDQ showed a hepatoprotective effect in CCl4-induced liver damage and was more effective than silymarin in relation to activity of ALT, AST, GGT, concentration of TG and histopathological score (P<0.05).
In order to understand the role of oxidative stress in the pathogenesis of toxic liver damage, we analyzed the effect of BHDQ on redox status in rats treated with CCl4. CCl4 appeared to cause an increase in Imax (P<0.001) and S (P<0.001), reflecting the intensity of free radical oxidation in the liver and blood serum, compared to animals in the control group (Table 1 and Table 2).
| Indicator | Control | CCl4 | CCl4+BHDQ 25 | CCl4+BHDQ 50 | BHDQ | CCl4+silymarin |
| Imax (mV) | 42.03±8.41 | 86.72±17.29*** | 44.02±6.21## | 39.04±5.37### | 33.62±6.72* | 63.58±12.71##,&& |
| S (mV*s) | 92.32±4.61 | 236.04±11.82*** | 120.88±24.23## | 98.71±19.69### | 91.37±18.32 | 112.06±22.41## |
| tgα2 | 8.11±1.60 | 16.70±3.31*** | 10.22±2.28## | 8.91±1.80### | 6.51±1.33 | 14.11±2.80#,&& |
| DC (µmol/mL homogenate) | 9.01±1.80 | 15.71±3.14*** | 8.47±1.72### | 10.20±2.00### | 9.91±2.02 | 13.03±2.63##,& |
| AH (U/g tissue) | 0.249±0.062 | 0.083±0.021*** | 0.177±0.043##,& | 0.207±0.052### | 0.224±0.056 | 0.150±0.038###,&& |
| Citrate (µmol/mL homogenate) | 0.65±0.13 | 1.31±0.26*** | 0.84±0.17##,& | 0.64±0.13### | 0.70±0.14 | 0.75±0.15##,&& |
| Data are presented as mean±SD (n=10 in each group). Statistical analysis was performed using one-way ANOVA with Tukey's post hoc test. *P<0.05 and ***P<0.001 vs. the control group; #P<0.05, ##P<0.01, and ###P<0.001 vs. the CCl4 group; &P<0.05 and &&P<0.01 vs. the CCl4+BHDQ 50 group. Imax: maximum intensity of chemiluminescence; S: light sum of chemiluminescence; tgα2: tangent of the biochemiluminescence kinetic curve slope; DC: diene conjugates; AH: aconitate hydratase. | ||||||
| Indicator | Control | CCl4 | CCl4+BHDQ 25 | CCl4+BHDQ 50 | BHDQ | CCl4+silymarin |
| Imax (mV) | 25.22±6.31 | 51.20±12.78*** | 35.10±8.83###,& | 21.71±5.40### | 23.56±5.87 | 28.00±7.03###,& |
| S (mV*s) | 307.70±76.91 | 739.79±184.92*** | 305.04±76.30### | 317.22±79.31### | 292.33±73.08 | 314.11±78.52### |
| tgα2 | 13.30±2.72 | 29.51±5.93*** | 15.04±2.76### | 13.41±3.41### | 11.31±2.3 | 15.40±3.13###,& |
| DC (µmol/mL) | 7.50±1.51 | 16.04±3.22*** | 8.95±1.81### | 10.42±2.11### | 7.13±1.41 | 9.90±2.00### |
| AH (U/mL) | 0.061±0.012 | 0.025±0.005*** | 0.029±0.003& | 0.061±0.015### | 0.067±0.013 | 0.061±0.012### |
| Citrate (mmol/L) | 0.57±0.11 | 1.24±0.25*** | 1.17±0.23& | 0.61±0.11### | 0.54±0.10 | 0.77±0.15##,& |
| Data are presented as mean±SD (n=10 in each group). Statistical analysis was performed using one-way ANOVA with Tukey's post hoc test. ***P<0.001 vs. the control group; ##P<0.01 and ###P<0.001 vs. the CCl4 group; &P<0.05 vs. the CCl4+BHDQ 50 group. Imax: maximum intensity of chemiluminescence; S: light sum of chemiluminescence; tgα2: tangent of the biochemiluminescence kinetic curve slope; DC: diene conjugates; AH: aconitate hydratase. | ||||||
Total antioxidant activity in the liver and serum also increased which was evidenced by an increase in tgα2 (P<0.001) (Table 1 and Table 2). In addition, rats with CCl4-induced liver damage were characterized by an increased level of primary products of lipid peroxidation (i.e., DC) (P<0.001), decreased activity of AH (P<0.001), sensitive to ROS, and accumulation of AH substrate citrate (P<0.001), compared to the control group. BHDQ at a dose of 50 mg/kg BW changed these parameters moving them closer to control values in contrast to animals of CCl4 group (all P<0.001), but strong evidence of greater efficacy for all dosages was not obtained (Table 1 and Table 2). Data indicate that BHDQ exhibited hepatoprotective activity due to the presence of an antioxidant effect.
Silymarin also contributed to a significant change in all analyzed parameters in rats with CCl4-induced liver damage (all P<0.001). However, this was less effective in relation to Imax, tgα2 and AH activity, DC concentration in the liver, and citrate level compared to BHDQ at a dose of 50 mg/kg BW (P<0.001 for Imax, P=0.001 for tgα2, P=0.009 for AH activity, P=0.01 for DC concentration, and P=0.001 for citrate level) (Table 1 and Table 2).
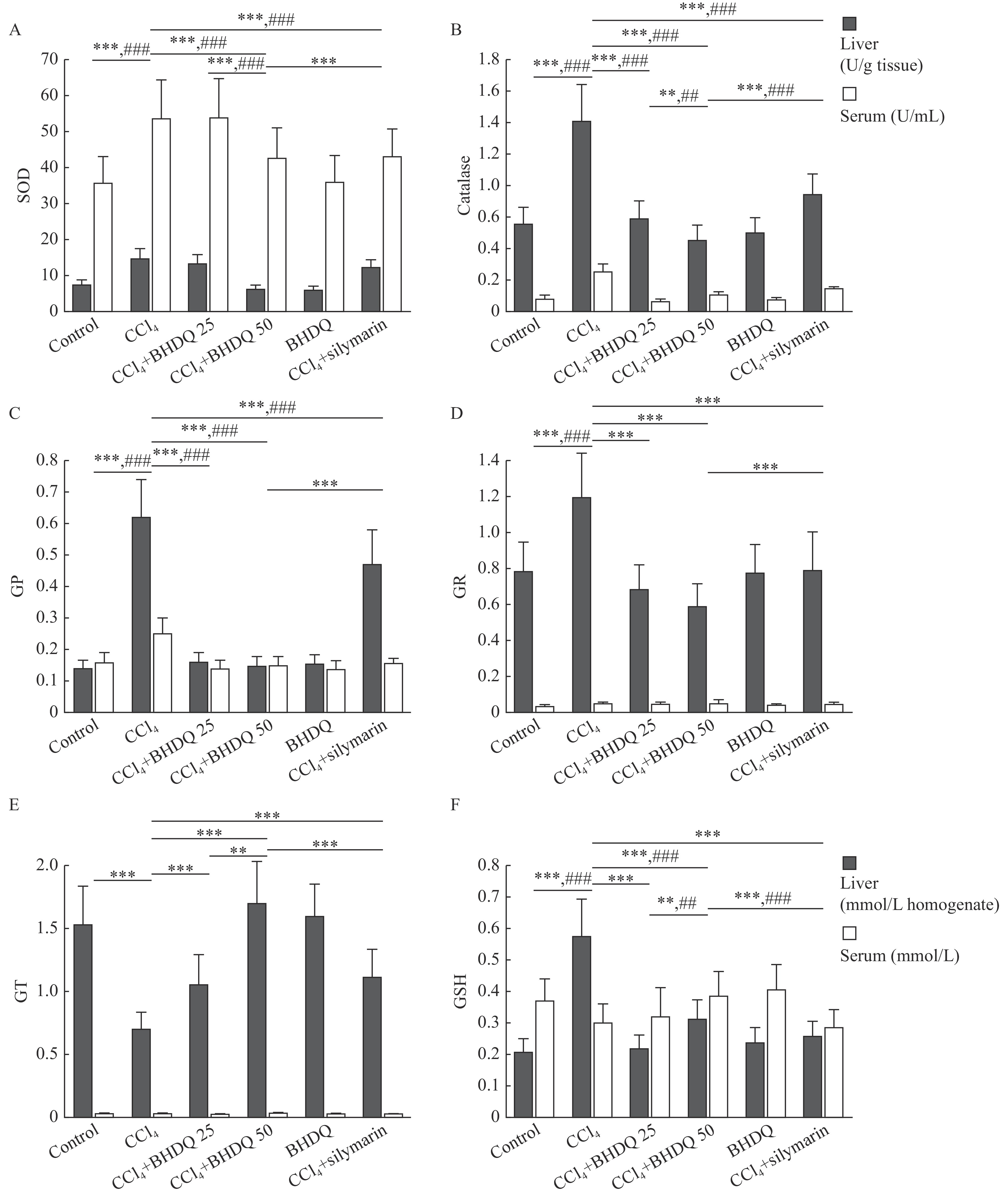
Protection against ROS formed during CCl4 metabolism is provided by an antioxidant system including enzymes such as SOD, catalase, GP, GR, and GT. The administration of CCl4 to laboratory animals was associated with a significant (all P<0.001) increase in the activity of SOD, catalase, GP and GR in the liver and blood serum compared to the control group (Fig. 4A–D). A decrease in GT activity in the liver (P<0.001) and a multi-directional change in the content of GSH in liver and blood serum (all P<0.001) were observed in animals with CCl4 group (Fig. 4E and F). In turn, BHDQ at a dose of 50 mg/kg BW contributed to a change in the activity of SOD, catalase, GP, GR, GT and GSH concentration on control values (all P<0.001) in rats with CCl4-induced liver injury.
For the activity of SOD (P<0.001), catalase (P=0.001), GT (P=0.001) and GSH concentration (P=0.003), more significant changes relative to BHDQ 25 group were observed for BHDQ at a dose of 50 mg/kg BW (Fig. 4A–F). Additionally, the effectiveness of silymarin was significantly lower (all P<0.001) based on the analysis of SOD, catalase, GP, GR, GT activity and GSH concentration compared with the CCl4+BHDQ 50 group, or with both groups of rodents with CCl4-induced liver injury receiving BHDQ (Fig. 4A–F).
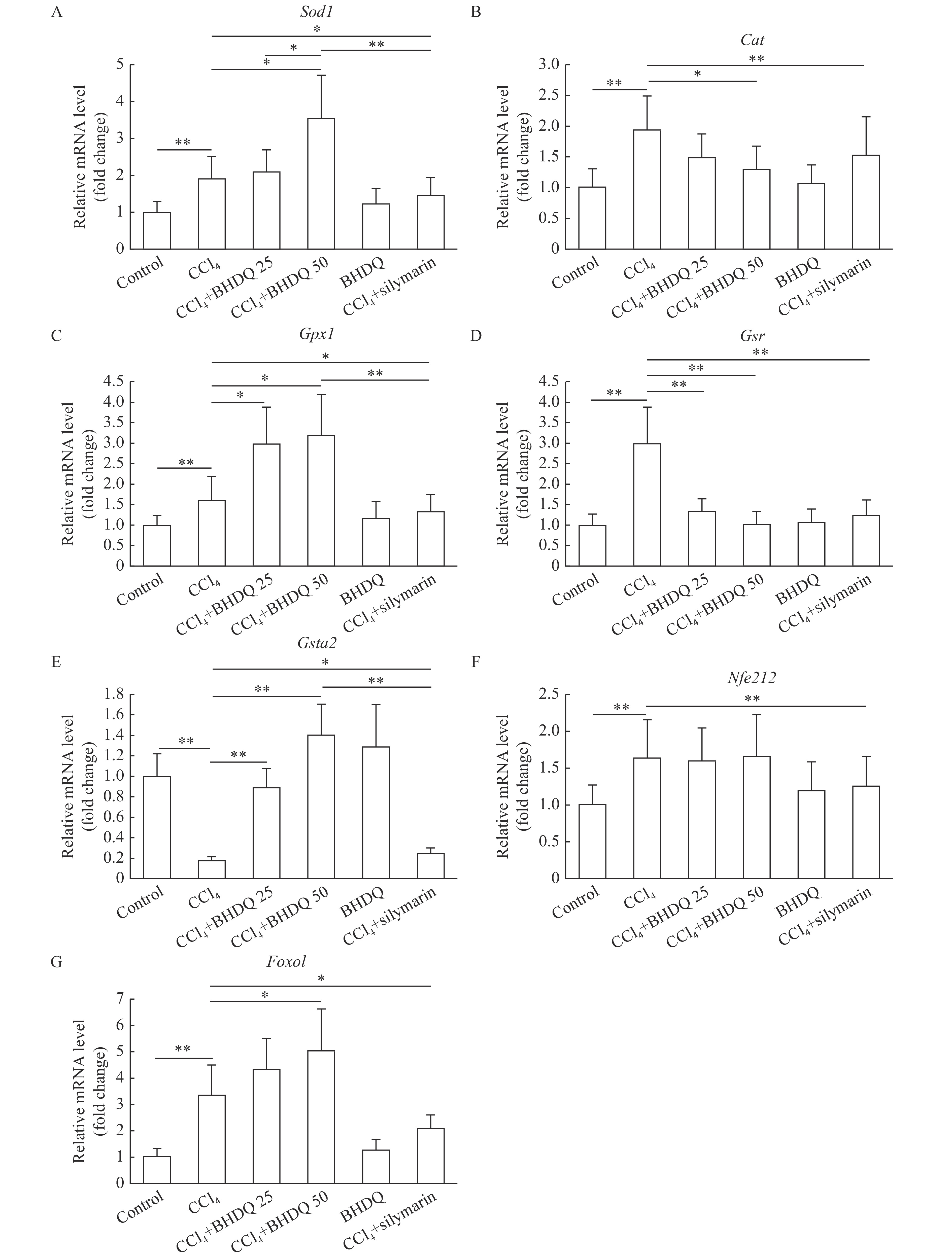
The induction of toxic hepatic injury in rats was associated with activated gene expression of Sod1 (P=0.001), Cat (P=0.001), Gpx1 (P=0.009), Gsr (P=0.001), Nfe2l2 (P=0.002) and Foxo1 (P=0.001) (Fig. 5A–D, F and G) compared to control group; however, a decrease in the level of mRNA of the Gsta2 (P<0.001) gene was observed (Fig. 5E). BHDQ at a dose of 50 mg/kg BW for rats with CCl4-induced hepatic injury was associated with additional activation of expression of Sod1 (P=0.01), Gpx1 (P=0.011) and Foxo1 (P=0.021) (Fig. 5A, C and G). The level of Cat (P=0.036), Gsr (P=0.002) and Gsta2 (P=0.003) transcripts changed toward control when BHDQ was administered to animals of CCl4 group (Fig. 5B, D and E). Whereas, silymarin changed in terms of Sod1, Cat, Gpx1, Gsr, Gsta2, Nfe2l2 and Foxo1 expression on values of control group (P<0.05).
BHDQ had a more significant modulatory effect on the functioning of the antioxidant system compared with silymarin.
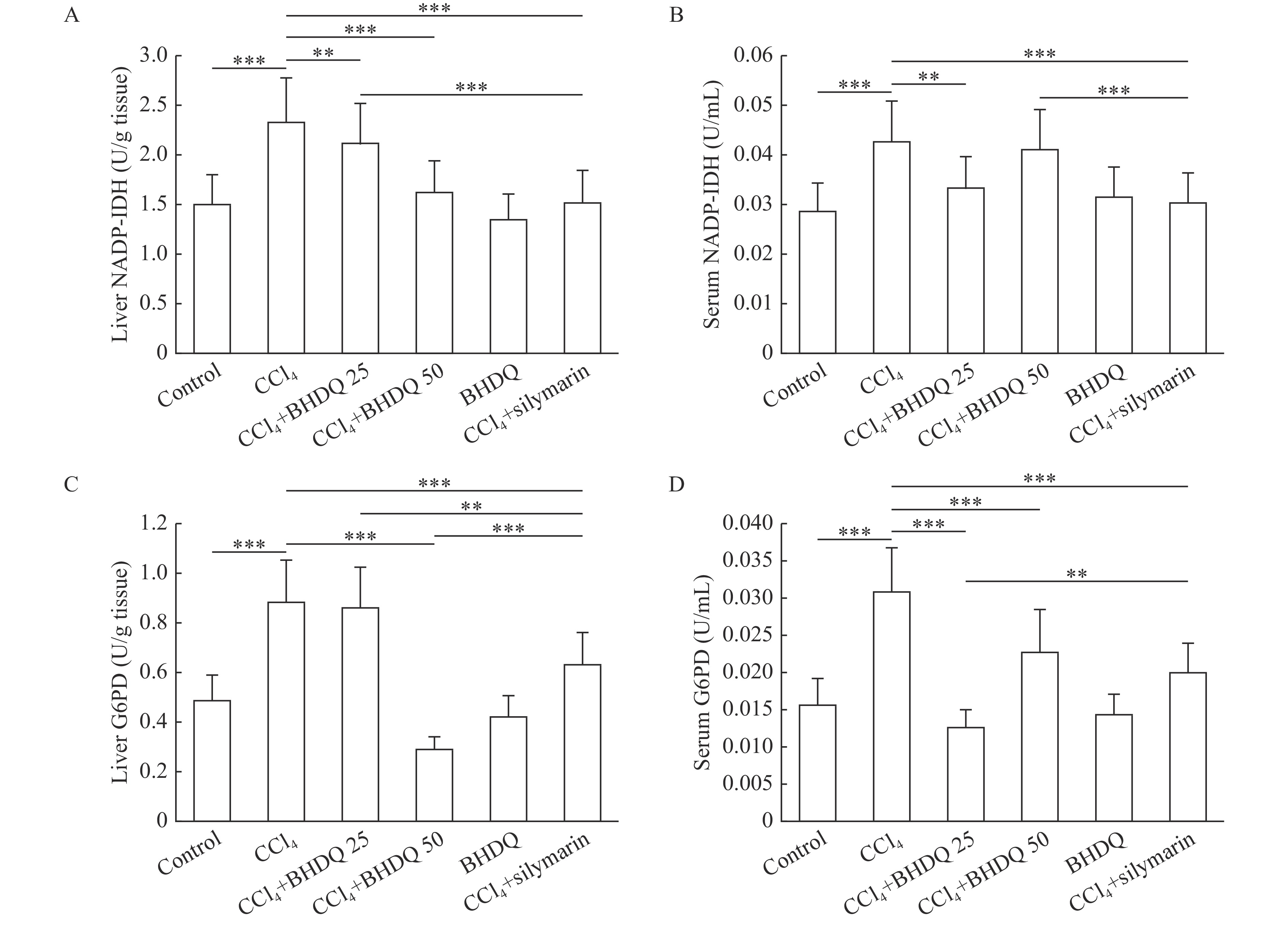
It was found that CCl4-induced hepatic injury in rats was associated with NADP-IDH (Fig. 6A and B) and G6PD (Fig. 6C and D) activation in the liver and blood serum compared to the control group (all P<0.001). BHDQ at a dose of 50 mg/kg BW contributed to the change of NADP-IDH and G6PD activity on the control values in liver of rats with CCl4-induced hepatic injury (all P<0.001). However, similar dose-dependencies were not observed when analyzing tissues. There was no unequivocal evidence for a more effective effect of both doses of BHDQ compared to silymarin on NADPH-supplying enzymes.
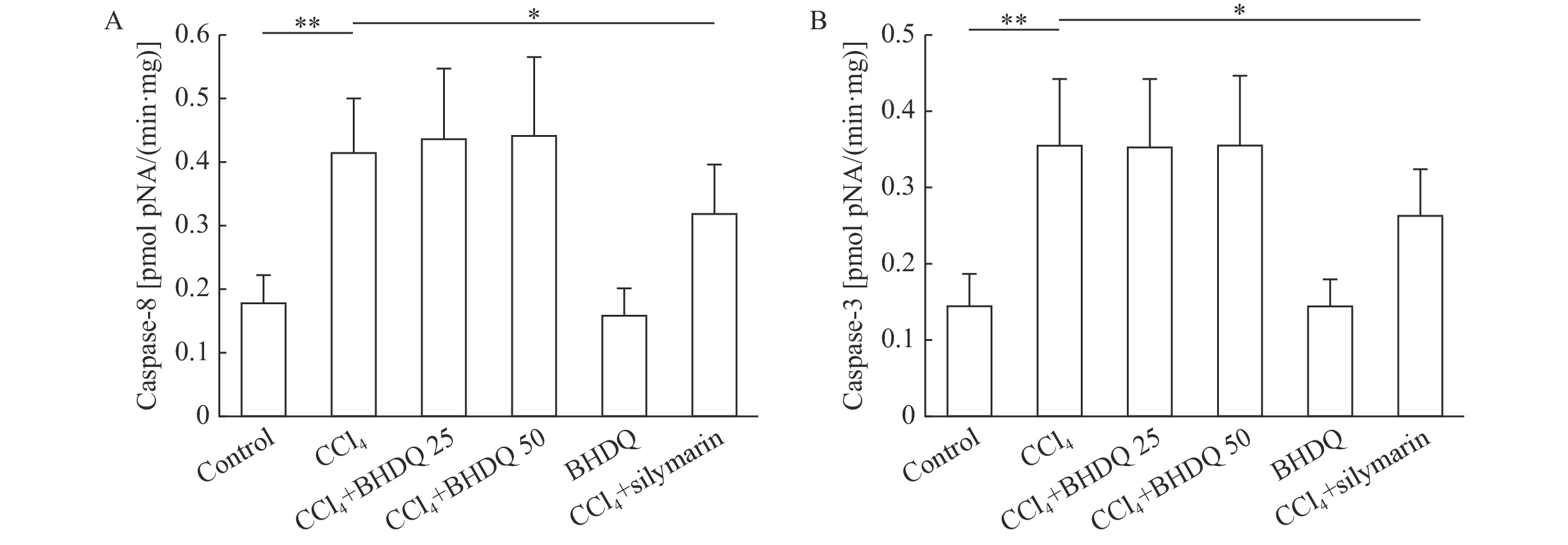
Oxidative stress and inflammation developing through CCl4-induced liver injury can initiate apoptosis by activating inducible and effector caspases. As this study has shown, CCl4-induced hepatic injury promoted the activation of inductor caspase-8 (P=0.001) (Fig. 7A) and effector caspase-3 (P=0.001) compared to the values of the control animals (Fig. 7B) (P<0.05). However, BHDQ did not significantly affect caspases activities while silymarin did (all P<0.05).
Oxidative stress impairs hepatic parenchyma cells, extracellular matrix, and immune activation, thus playing a central role in CCl4-induced hepatic injury[4]. It has previously been shown that the pronounced antioxidant effect of 6-hydroxy-2,2,4-trimethyl-1,2-dihydroquinoline reduces CCl4-induced hepatic injury[10]. However, it is also known that ethoxyquin, an antioxidant from the same series of compounds, has pro-oxidant as well as carcinogenic effects[21]. Here we structurally modified 6-hydroxy-2,2,4-trimethyl-1,2-dihydroquinoline by introducing a benzoyl group to the nitrogen atom. This was done to prevent the generation of the toxic metabolite 2,2,4-trimethyl-6(2H)-quinolinone[11].
We found in terms of hepatoprotectivity that BHDQ was related to a decreased level of oxidative stress, marker enzymes of hepatic cytolysis. We also observed improved morphologies in the liver tissue in rats treated with CCl4. Increased ROS production induced by CCl4 is known to play a role in the development of hepatic steatosis. Therefore, activation of Cyp2E1 by CCl4 leads to the formation of toxic peroxyl and alkoxyl radicals which initiate lipid peroxidation[22]. In turn, BHDQ reduced fatty liver infiltration and probably, BHDQ antioxidation became the key mechanism involved in hepatoprotectivity in regard to this compound. Thus, the reduction of oxidative stress and fatty infiltration of hepatocytes under the action of BHDQ appeared to be the key factor in the improvement of histopathological score of liver parenchyma and the reduction of hepatic enzyme activity in serum of rats with CCl4-induced hepatic injury.
The liver has its own antioxidant defence system against ROS. The main antioxidant enzymes include SOD, catalase, GP, GT, and GR, which catalyze the reduction of GSSG to GSH. NADPH is required for the reduction of GSSG in the GR-catalysed reaction which essentially constrains the limiting factor. The main suppliers of NADPH are NADP-IDH and G6PD[23]. We demonstrated an increase in the activity of the majority of the antioxidant enzymes in the CCl4 group. This was apparently the result of an adaptive response to the introduction of xenobiotics and the development of oxidative stress.
Along with this, CCl4 was associated with an imbalance in the antioxidant system, which was expressed as inhibited GT activity and in multidirectional changes related to GSH concentrations. BHDQ in rats with CCl4-induced liver damage contributed to the normalization of the analyzed. BHDQ appeared to exhibit antioxidant activity and improved the redox status in the liver of animals with CCl4-induced liver damage. Thus, the change in the activity of antioxidant enzymes could be the result of a decrease in their load during the reduction of ROS level by BHDQ.
One of the central regulators of the antioxidant system is the Nrf2 transcription factor which is encoded by the Nfe2l2 gene[24]. Oxidative stress also causes a significant increase in the transcriptional activity of Foxo1, and this affects the expression of SOD and catalase[25]. Our findings revealed an increase in mRNA levels of antioxidant enzyme genes in CCl4-dependent hepatic injury. However, a decrease in expression of Gsta2, which correlated with GT activity in the liver, was also revealed. BHDQ administration contributed to a decrease in Cat and Gsr transcript levels, and an increase in Gsta2 mRNA levels, relative to animals with CCl4-induced hepatic injury. We also found that there was additional activation of the Sod1, Gpx1 and Foxo1 genes in this study.
BHDQ appears to have a stimulating effect on the expression of genes encoding antioxidant enzymes. It can be assumed that BHDQ effect manifests by way of Nfe2l2 and Foxo1 and by transcriptional regulation. In particular, it is known that dihydroquinoline ethoxyquin is a selective activator of Nrf2[26]. Therefore we suggest, that despite the decrease in oxidative stress intensity and the normalization of antioxidant enzymes, the content of mRNA encoding for these enzymes still increased with BHDQ. This somewhat counter-intuitive idea is also associated with the apparent inducing effect which should be considered more closely. It appears that the normalization of SOD and GP activity could be related to other regulatory mechanisms, in particular a change in their catalytic properties.
In CCl4-induced damage, ROS attack various hepatocellular structures causing the release of proinflammatory mediators. In particular, tumor necrosis factor-α (TNF-α) is released, which contributes to the development of apoptosis. TNF-α activates the Fas ligand, which is followed by the generation of an apoptotic complex for binding and activation of procaspase-8. Caspase-8 can then activate effector caspase-3, which ultimately leads to cellular apoptosis[27]. In this study, we noticed an increase in caspase-8 and caspase-3 activity after CCl4. BHDQ had no effect on the activity of these apoptosis mediating enzymes. It is likely that the limiting factor was duration of the experiment which was four days from CCl4 administration. This was most likely insufficient to track the effect of BHDQ on caspase activity.
In summary, we did not observe BHDQ dose-dependency in relation to oxidative stress or other indicators. There was more pronounced hepatoprotectivity in relation to this compound at a dose of 25 mg/kg BW. This activity does appear to be superior to silymarin, although this was a relatively small rodent modelling study. We can conclude that further BHDQ studies are warranted although we must pay particular attention to dose-responses. Providing BHDQ to rats with CCl4-induced hepatic injury had a hepatoprotective effect which is associated with the antioxidation capacity of the tested compound.
This work was supported by the Russian Foundation for Basic Research (Grant No. 20-04-00526 А).
CLC number: R965, Document code: A
The authors reported no conflict of interests.
| [1] |
Wang R, Tang R, Li B, et al. Gut microbiome, liver immunology, and liver diseases[J]. Cell Mol Immunol, 2021, 18(1): 4–17. doi: 10.1038/s41423-020-00592-6
|
| [2] |
Lee YS, Cho IJ, Kim JW, et al. Hepatoprotective effects of blue honeysuckle on CCl4-induced acute liver damaged mice[J]. Food Sci Nutr, 2019, 7(1): 322–338. doi: 10.1002/fsn3.893
|
| [3] |
Saijou E, Enomoto Y, Matsuda M, et al. Neutrophils alleviate fibrosis in the CCl4-induced mouse chronic liver injury model[J]. Hepatol Commun, 2018, 2(6): 703–717. doi: 10.1002/hep4.1178
|
| [4] |
Dutta S, Chakraborty AK, Dey P, et al. Amelioration of CCl4 induced liver injury in swiss albino mice by antioxidant rich leaf extract of Croton bonplandianus Baill[J]. PLoS One, 2018, 13(4): e0196411. doi: 10.1371/journal.pone.0196411
|
| [5] |
Mortezaee K, Khanlarkhani N. Melatonin application in targeting oxidative-induced liver injuries: a review[J]. J Cell Physiol, 2018, 233(5): 4015–4032. doi: 10.1002/jcp.26209
|
| [6] |
Di Costanzo A, Angelico R. Formulation strategies for enhancing the bioavailability of silymarin: the state of the art[J]. Molecules, 2019, 24(11): 2155. doi: 10.3390/molecules24112155
|
| [7] |
Tereshchenko OG, Nikolskaya ED, Zhunina OA, et al. Formulation of perspective hepatoprotector polymeric forms based on silybin and ursodeoxycholic acid[J]. Russ Chem Bull, 2018, 67(12): 2290–2296. doi: 10.1007/s11172-018-2372-4
|
| [8] |
Muriel P, Rivera-Espinoza Y. Beneficial drugs for liver diseases[J]. J Appl Toxicol, 2008, 28(2): 93–103. doi: 10.1002/jat.1310
|
| [9] |
Yu Y, Cai J, She Z, et al. Insights into the epidemiology, pathogenesis, and therapeutics of nonalcoholic fatty liver diseases[J]. Adv Sci (Weinh), 2019, 6(4): 1801585. doi: 10.1002/advs.201801585
|
| [10] |
Iskusnykh IY, Kryl'skii ED, Brazhnikova DA, et al. Novel antioxidant, deethylated ethoxyquin, protects against carbon tetrachloride induced hepatotoxicity in rats by inhibiting NLRP3 inflammasome activation and apoptosis[J]. Antioxidants (Basel), 2021, 10(1): 122. doi: 10.3390/antiox10010122
|
| [11] |
Kryl'skii ED, Chupandina EE, Popova TN, et al. 1-benzoyl-6-hydroxy-2,2,4-trimethyl-1,2-dihydroquinoline exerts a neuroprotective effect and normalises redox homeostasis in a rat model of cerebral ischemia/reperfusion[J]. Metab Brain Dis, 2022, 37(4): 1271–1282. doi: 10.1007/s11011-022-00928-3
|
| [12] |
Onyibe PN, Edo GI, Nwosu LC, et al. Effects of vernonia amygdalina fractionate on glutathione reductase and glutathione-S-transferase on alloxan induced diabetes wistar rat[J]. Biocatal Agric Biotechnol, 2021, 36: 102118. doi: 10.1016/j.bcab.2021.102118
|
| [13] |
Knodell RG, Ishak KG, Black WC, et al. Formulation and application of a numerical scoring system for assessing histological activity in asymptomatic chronic active hepatitis[J]. Hepatology, 1981, 1(5): 431–435. doi: 10.1002/hep.1840010511
|
| [14] |
Piskarev IM, Trofimova SV, Burkhina OE, et al. Investigation of the level of free-radical processes in substrates and biological samples using induced chemiluminescence[J]. Biophysics, 2015, 60(3): 400–408. doi: 10.1134/S0006350915030148
|
| [15] |
Guilbault GG. Handbook of enzymatic methods of analysis[M]. New York: Marcel Dekker, 1976: 752.
|
| [16] |
Rao UM. Source of superoxide anion radical in aerobic mixtures consisting of NADH, 5-methylphenazinium methyl sulfate and nitroblue tetrazolium chloride[J]. Free Radical Biol Med, 1989, 7(5): 513–519. doi: 10.1016/0891-5849(89)90027-0
|
| [17] |
Góth L. A simple method for determination of serum catalase activity and revision of reference range[J]. Clin Chim Acta, 1991, 196(2–3): 143–151. doi: 10.1016/0009-8981(91)90067-m
|
| [18] |
Paglia DE, Valentine WN. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase[J]. J Lab Clin Med, 1967, 70(1): 158–169. https://www.translationalres.com/article/0022-2143(67)90076-5/pdf
|
| [19] |
Zanetti G. Rabbit liver glutathione reductase. Purification and properties[J]. Arch Biochem Biophys, 1979, 198(1): 241–246. doi: 10.1016/0003-9861(79)90415-6
|
| [20] |
Warholm M, Guthenberg C, von Bahr C, et al. Glutathione transferases from human liver[J]. Methods Enzymol, 1985, 113: 499–504. doi: 10.1016/s0076-6879(85)13065-x
|
| [21] |
Błaszczyk A, Augustyniak A, Skolimowski J. Ethoxyquin: an antioxidant used in animal feed[J]. Int J Food Sci, 2013, 2013: 585931. doi: 10.1155/2013/585931
|
| [22] |
Jin C, Li B, Lin S, et al. Mechanism of the inhibitory effects of Eucommia ulmoides oliv. cortex extracts (EUCE) in the CCl4-induced acute liver lipid accumulation in rats[J]. Int J Endocrinol, 2013, 2013: 751854. doi: 10.1155/2013/751854
|
| [23] |
Popov SS, Anufrieva EI, Kryl'skii ED, et al. The effect of methylethylpiridinol addition to the therapy on the level of pigment epithelium-derived factor and oxidative status in patients with diabetic nephropathy: randomized controlled open-label clinical study[J]. J Diabetes Metab Disord, 2021, 20(1): 709–717. doi: 10.1007/s40200-021-00802-6
|
| [24] |
Zhuang Y, Wu H, Wang X, et al. Resveratrol attenuates oxidative stress-induced intestinal barrier injury through PI3K/Akt-mediated Nrf2 signaling pathway[J]. Oxid Med Cell Longev, 2019, 2019: 7591840. doi: 10.1155/2019/7591840
|
| [25] |
Xing Y, Li A, Yang Y, et al. The regulation of FOXO1 and its role in disease progression[J]. Life Sci, 2018, 193: 124–131. doi: 10.1016/j.lfs.2017.11.030
|
| [26] |
Miao W, Hu L, Kandouz M, et al. A cell-based system to identify and characterize the molecular mechanism of drug-metabolizing enzyme (DME) modulators[J]. Biochem Pharmacol, 2004, 67(10): 1897–1905. doi: 10.1016/j.bcp.2004.02.009
|
| [27] |
Xu G, Han X, Yuan G, et al. Screening for the protective effect target of deproteinized extract of calf blood and its mechanisms in mice with CCl4-induced acute liver injury[J]. PLoS One, 2017, 12(7): e0180899. doi: 10.1371/journal.pone.0180899
|
| Indicator | Control | CCl4 | CCl4+BHDQ 25 | CCl4+BHDQ 50 | BHDQ | CCl4+silymarin |
| Imax (mV) | 42.03±8.41 | 86.72±17.29*** | 44.02±6.21## | 39.04±5.37### | 33.62±6.72* | 63.58±12.71##,&& |
| S (mV*s) | 92.32±4.61 | 236.04±11.82*** | 120.88±24.23## | 98.71±19.69### | 91.37±18.32 | 112.06±22.41## |
| tgα2 | 8.11±1.60 | 16.70±3.31*** | 10.22±2.28## | 8.91±1.80### | 6.51±1.33 | 14.11±2.80#,&& |
| DC (µmol/mL homogenate) | 9.01±1.80 | 15.71±3.14*** | 8.47±1.72### | 10.20±2.00### | 9.91±2.02 | 13.03±2.63##,& |
| AH (U/g tissue) | 0.249±0.062 | 0.083±0.021*** | 0.177±0.043##,& | 0.207±0.052### | 0.224±0.056 | 0.150±0.038###,&& |
| Citrate (µmol/mL homogenate) | 0.65±0.13 | 1.31±0.26*** | 0.84±0.17##,& | 0.64±0.13### | 0.70±0.14 | 0.75±0.15##,&& |
| Data are presented as mean±SD (n=10 in each group). Statistical analysis was performed using one-way ANOVA with Tukey's post hoc test. *P<0.05 and ***P<0.001 vs. the control group; #P<0.05, ##P<0.01, and ###P<0.001 vs. the CCl4 group; &P<0.05 and &&P<0.01 vs. the CCl4+BHDQ 50 group. Imax: maximum intensity of chemiluminescence; S: light sum of chemiluminescence; tgα2: tangent of the biochemiluminescence kinetic curve slope; DC: diene conjugates; AH: aconitate hydratase. | ||||||
| Indicator | Control | CCl4 | CCl4+BHDQ 25 | CCl4+BHDQ 50 | BHDQ | CCl4+silymarin |
| Imax (mV) | 25.22±6.31 | 51.20±12.78*** | 35.10±8.83###,& | 21.71±5.40### | 23.56±5.87 | 28.00±7.03###,& |
| S (mV*s) | 307.70±76.91 | 739.79±184.92*** | 305.04±76.30### | 317.22±79.31### | 292.33±73.08 | 314.11±78.52### |
| tgα2 | 13.30±2.72 | 29.51±5.93*** | 15.04±2.76### | 13.41±3.41### | 11.31±2.3 | 15.40±3.13###,& |
| DC (µmol/mL) | 7.50±1.51 | 16.04±3.22*** | 8.95±1.81### | 10.42±2.11### | 7.13±1.41 | 9.90±2.00### |
| AH (U/mL) | 0.061±0.012 | 0.025±0.005*** | 0.029±0.003& | 0.061±0.015### | 0.067±0.013 | 0.061±0.012### |
| Citrate (mmol/L) | 0.57±0.11 | 1.24±0.25*** | 1.17±0.23& | 0.61±0.11### | 0.54±0.10 | 0.77±0.15##,& |
| Data are presented as mean±SD (n=10 in each group). Statistical analysis was performed using one-way ANOVA with Tukey's post hoc test. ***P<0.001 vs. the control group; ##P<0.01 and ###P<0.001 vs. the CCl4 group; &P<0.05 vs. the CCl4+BHDQ 50 group. Imax: maximum intensity of chemiluminescence; S: light sum of chemiluminescence; tgα2: tangent of the biochemiluminescence kinetic curve slope; DC: diene conjugates; AH: aconitate hydratase. | ||||||