| Citation: | Zhang Jiarong, Sun Hui, Pei Wei, Jiang Huijun, Chen Jin. Nanobody-based immunosensing methods for safeguarding public health[J]. The Journal of Biomedical Research, 2021, 35(4): 318-326. DOI: 10.7555/JBR.35.20210108 |
In principle, an immunosensing method is a biosensing strategy based on specific recognition between an antigen and an antibody, which measures the signals of a broad biological target by a concentration-dependent approach[1]. Thus far, the reported immunosensing methods mainly utilize enzyme-linked immunosorbent assays, photo-/electrochemical immunosensors, and chemiluminescence assays. Compared with other established analytical methods, such as chromatographic and mass spectrometric analysis, an immunosensing method offers many advantages including high sensitivity, portability, rapid rate, and miniaturization potential for point-of-care testing[2–6]. In particular, immunosensors employ certain antibodies immobilized on an electrode surface as molecular probes to directly capture target molecules for signal read-out, and have witnessed a rapid growth in many aspects including diseases diagnosis[7–9], environmental pollution monitoring[10–12], and pathogenic microorganism detection[5,13].
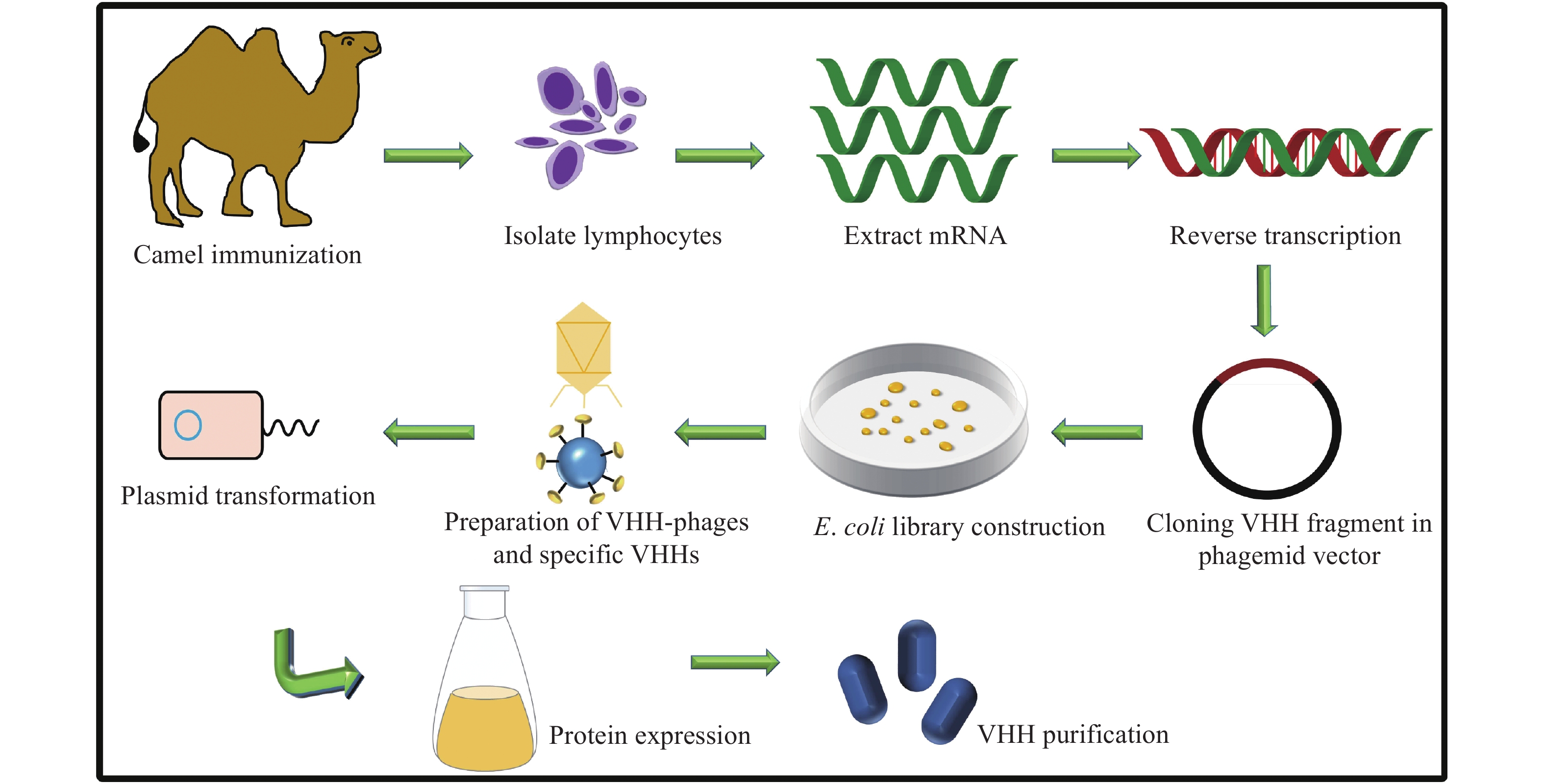
It is known that in immunosensing methods, the stably formed immunocomplex between the targeted antigen and antibody (Ka=1010 M−1) on the biosensing interface is responsible for their high sensitivity and selectivity[14]. A single-domain nanobody consists of an antigen-binding variable region, which is different from the traditional antibodies, and it achieves versatile biomedical applications owing to its retained cognate antigen-binding specificity, unique compact structure, and excellent physicochemical properties[15–16]. Historically, after Hamers-Casterman et al first identified a heavy-chain-only antibody from the Camelidae family in 1993, the term nanobody originated from its nanosized structure[17], which was formed by an autonomous single variable region, also called as VHH[18]. As shown in Fig. 1, the biotech production of an antigen-specific nanobody is as follows[19]. Lymphocytes are collected from an immunized camel, to extract the mRNA. Subsequently, the cDNA is synthesized and packed into a phagemid. Following, the phage library is constructed for the selection of the antigen-specific nanobody. Finally, the nanobody-encoded gene is subcloned into an expression vector to produce a soluble nanobody.
Several attributes of a nanobody are beneficial for the novel design and practices of immunosensing methods. In conventional full-length immunoglobulin G, hypervariable loops in the variable domain form a paratope complementary to the epitope on the antigen, which are referred to as complementary determining regions (CDRs) 1, 2, and 3[20]. In comparison, in a nanobody, the CDR3 loops are relatively longer[21], which enable more efficient contact with the buried surface area on the antigen, which may be inaccessible by conventional antibodies. Moreover, a nanobody is highly soluble with excellent physicochemical stability[22], and disulphide bond formation and glycosylation are not required for a nanobody to retain its antigen-binding properties[14], making the scale-up production of nanobodies of a recombinant form feasible[23]. Additionally, a nanobody populates in a minimal size of approximately 15 kDa, which results in a shorter circulatory half-life and improved penetration ability than the full-sized antibody[24].
There are many studies and related reviews on immunosensing methods, such as immunosensors. In this minireview, we do not aim to provide a comprehensive review about the progress in this field, instead, we mainly focus on the recent reports related to public health applications of a nanobody: clinical diagnosis, detection of pathogenic organisms, and environmental pollutions.
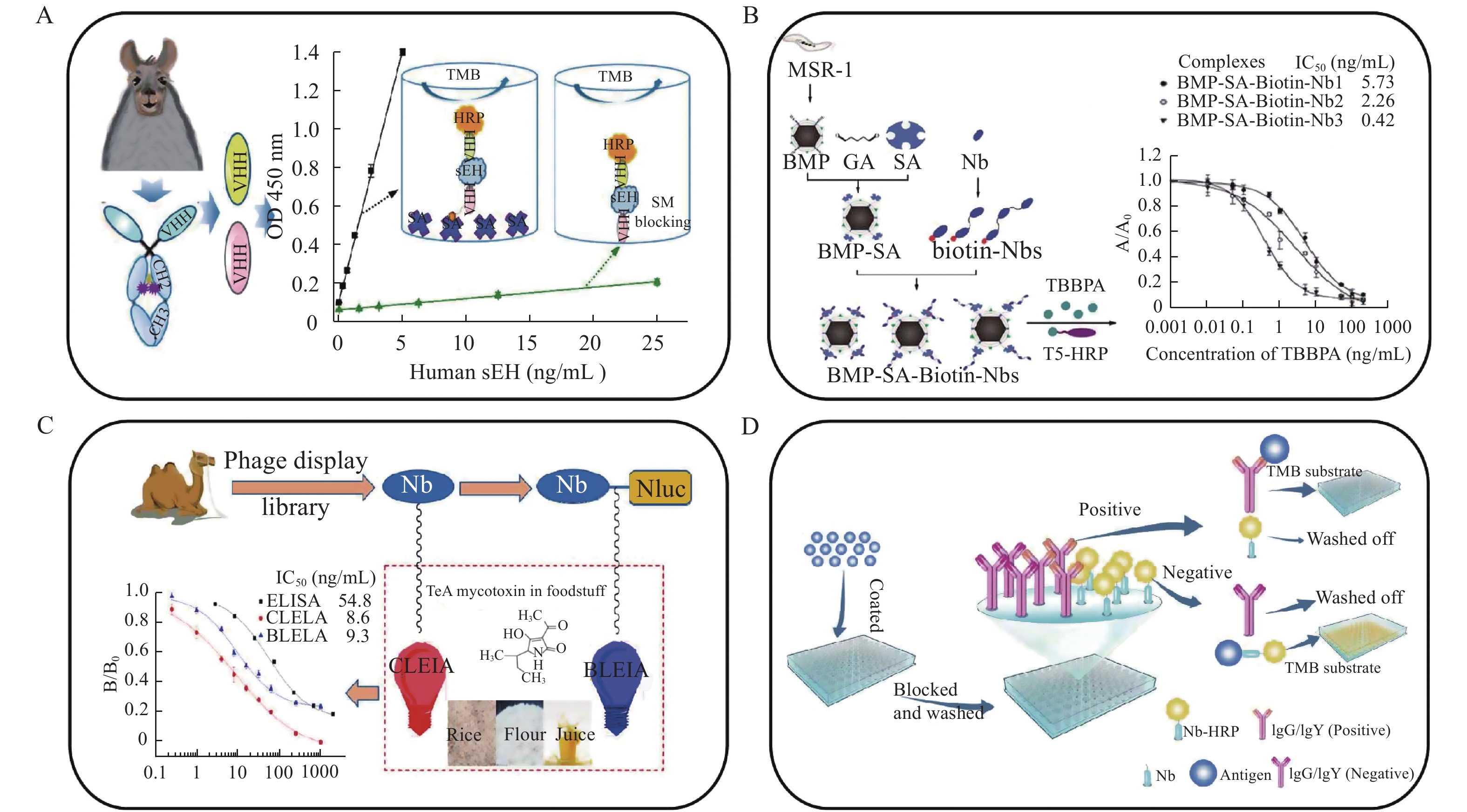
Owing to its operational convenience and cost-effectiveness, for decades, the enzyme-linked immunosorbent assay (ELISA) has been one of the most commonly used diagnostic methods both in clinics and laboratories to detect target antigens in biological systems[25]. It is based on specific antigen–antibody interactions. The analytical performance of ELISA is closely related to the affinity, specificity, and availability of the antibody used. However, the variance between batches of polyclonal antibodies and the deterioration of the hybridomas of monoclonal antibodies in storage have raised serious concerns regarding the reproducibility of the ELISA kit[26]. Additionally, traditional antibodies are glycosylated proteins, their production is time-consuming, and they are difficult to be massively expressed in heterologous systems[27]. By contrast, a nanobody can undergo genetic modification, which can be easily achieved in a prokaryotic system. Hammock et al reported a sandwich immunoassay based on a double-nanobody strategy (Fig. 2A) to determine human-soluble epoxide hydrolase, a biomarker of metabolic, cardiovascular, and chronic kidney diseases[26]. In the assay, nanobody A9 was first used as the capture antibody. Because passive adsorption of the antibody to polystyrene microplates may lead to loss of its binding capability, the capture nanobody was efficiently immobilized on the plates through streptavidin–biotin linkages. Subsequently, horseradish peroxidase (HRP)-labelled nanobody A1 synthesized via periodate oxidation (Maraprade reaction) was used as the detection antibody. The constructed ELISA kit achieved high sensitivity and a low limit of detection (LOD) (0.03 ng/mL), and was successfully applied to human tissue samples with good recovery and negligible cross-reactivity. The study demonstrated a good prototype of a nanobody-based ELISA kit for various targeted biological molecules.
In addition to disease-related biomarkers, a nanobody-based immunoassay can be used for monitoring environmental pollution. As shown in Fig. 2B, Xu et al developed three types of nanobody-based ELISA—monovalent (Nb1), bivalent (Nb2), and trivalent (Nb3)—to evaluate tetrabromobisphenol-A, in which the nanobody was conjugated with bacterial magnetic particles-streptavidin-biotin-Nbs (BMP-SA-Biotin-Nbs)[28]. It was found that the trivalent nanobody having high binding capability exhibited improved analytical performance. In addition, BMP-SA-Biotin-Nbs possessed high resistance to harsh conditions, such as high temperature, methanol, pH, and ionic strength, which is beneficial for its applications and storage. The BMP-SA-Biotin-Nb3-based assay possessed a linear range of 0.11–2.1 ng/mL with an LOD of 0.03 ng/mL. The determination results were consistent with those of liquid chromatography–tandem mass spectrometry, whereas the 30-minute assay time of the nanobody-based ELISA was relatively shorter than that of the mass spectrometric analysis. Furthermore, BMP-SA-Biotin-Nbs could be reused for thrice without apparent loss of the binding capability of the nanobodies.
Foodborne mycotoxin has posed serious threat to public health. To monitor the contamination of mycotoxins, two types of nanobody-based sandwich immunoassays—chemiluminescent enzyme immunoassay (CLEIA) and bioluminescent enzyme immunoassay (BLEIA)—have been developed (Fig. 2C)[29]. In the CLEIA, a common monovalent nanobody acted as the capture antibody, whereas a nanoluciferase-fused nanobody was employed in the BLEIA. For the determination of tenuazonic acid, both assays showed high sensitivity, satisfactory recoveries, and selectivity with LODs of 0.3 and 1.1 ng/mL for the CLEIA and the BLEIA, respectively.
To detect the Newcastle disease virus, an avian virus that has caused severe economic loss in the poultry industry worldwide[30], Zhao et al established a nanobody-based competitive enzyme-linked immunosorbent assay (cELISA), in which HRP was fused with a nanobody (Fig. 2D)[31]. The cut-off value of the cELISA was 18%, and its sensitivity and specificity were 100% and 98.6%, respectively, which were higher than those of the hemagglutination inhibition test and the commercial ELISA kit. Therefore, the proposed method in combination with the above-mentioned nanobody-based ELISA assays provided an advantageous solution for detecting biologically important molecules.
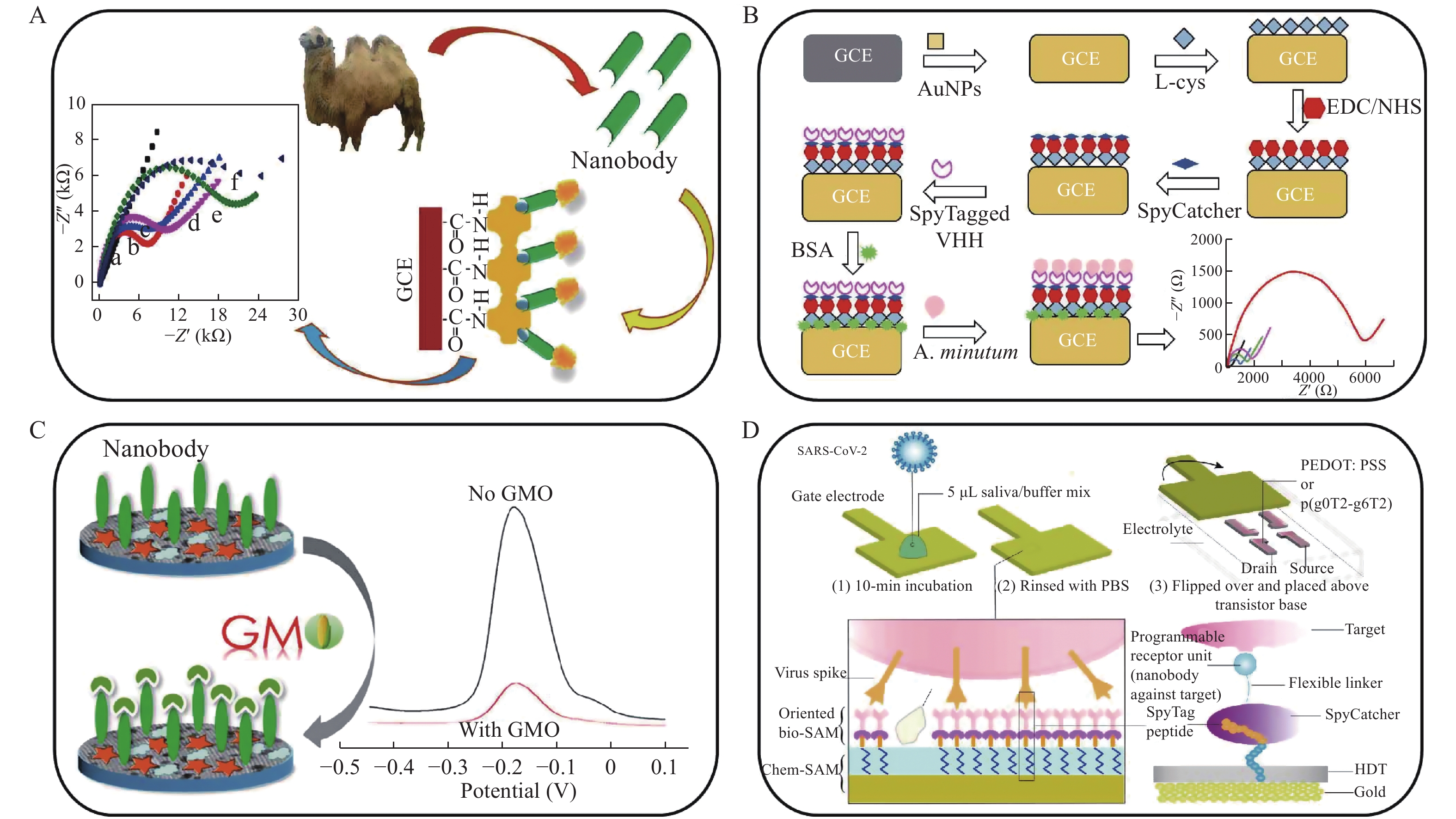
There are inherent disadvantages in enzyme-linked immunosorbent assays, such as dissatisfactory sensitivity. An electrochemical immunosensor is a powerful analytical method for the quantification of target molecules, which can provide rapid and accurate detection results owing to the well separation of the biorecognition and the signal output. An electrochemical immunosensing system is composed of biorecognition molecules, transduction elements, and read-out equipment. The biorecognition element, which is typically a capture antibody or antigen, is immobilized on an electrode interface, and the chemical information of an analyte, such as concentration, is detected and transformed into electric signals for measurement[32]. Wan et al reported nanobody-based electrochemical impedance spectroscopy for sensitive detection of testosterone, a biomarker of cardiac function and many diseases (Fig. 3A)[33]. The nanobody was immobilized on a glassy carbon electrode (GCE) via biotin and streptavidin linkages. The resulting calibration curve showed a wide linear range from 0.05 to 5 ng/mL with an LOD of 0.045 ng/mL.
Alexandrium minutum as a typical environmental pollutant has a progressively negative impact on the ecological environment. To deal with this problem, Ario de Marco et al established a nanobody-functionalized electrochemical immunosensor for monitoring the toxic microalgae (Fig. 3B)[34]. Desirable Alexandrium minutum detection results were achieved with the excellent design of a nanobody-immobilized sensing interface on a GCE. Specifically, first the electrode was modified with Au nanoparticles conjugated with L-cysteine to obtain a self-assembled monolayer. An Alexandrium minutum specific-nanobody was fused with SpyTag (a peptide isolated from Streptococcus pyogenes, which can bind to a protein partner via an amide bond in a few minutes[35]) and subsequently immobilized on a GCE modified with SpyCatcher (a protein partner). Charge transfer resistance changes were recorded using electrochemical impedance spectroscopy to quantify the Alexandrium minutum cells. In addition to a wide linear range of the calibration curve of Alexandrium minutum (103–109 cells/L) with an LOD of 3×103 cells/L, the developed immunoassay displayed high sensitivity and reproductivity, which are useful for the determination of environmental pollutants.
Genetically modified crops have generated considerable concerns regarding food safety. Shen et al constructed an electrochemical immunosensor combined with a nanobody to monitor a genetically modified crop using biomarker Agrobacterium sp. strain CP4 protein (CP4-EPSPS) (Fig. 3C)[36]. In the assay, an ordered mesoporous carbon having excellent thermal/mechanical stability and conductivity and Au nanoparticles were modified on a GCE to attach a redox probe, thionine (Th). As a capture agent, the anti-CP4-EPSPS nanobody was covalently bound to the modified GCE via a reaction between 1-ethyl-3-(3-(dimethylamino) propyl) carbodiimide (EDC) and N-hydroxysuccinimide (NHS). CP4-EPSPS could be detected with high sensitivity and specificity. The resulting calibration curve showed a linear range from 0.001 to 100 ng/mL with an LOD of 0.72 pg/mL, which is potentially applicable for screening genetically modified crops.
The pandemic of severe acute respiratory syndrome coronavirus 2 (also called COVID-19) has caused the death of millions of people and huge economic losses worldwide[37–38]. To retard the transmission of the infection, molecular diagnosis of the virus with high sensitivity is crucial. Sahika et al developed nanobody-functionalized organic electrochemical transistors for the detection of COVID-19 (Fig. 3D)[39]. For the detection of Middle East respiratory syndrome coronavirus, a spike-specific nanobody fused with the SpyCatcher protein was immobilized on an electrode interface through a SpyTag/SpyCatcher linkage. The overall assay could be completed within 15 minutes using 5 µL of the samples with a wide detection range from attomolar to nanomolar and an LOD of 23 fmol/L. The sensitivity of the proposed method was comparable to those of commercial ones; however, the assay time was relatively shorter. Thus, an electrochemical immunosensor provides a promising approach for rapid and sensitive detection of various pathogenic and environmental microbes.
A photoelectrochemical (PEC) immunosensor includes a photoelectric conversion unit in which immobilized photoactive materials are light-excited and the produced charges are transferred for the detection of target molecule[40]. Therefore, in addition to the specific recognition of immunocomplexes, a PEC immunosensor enables detection with a low interference background, excellent sensitivity, a rapid response, miniaturized equipment, and photoelectrochemical technique[41]. Typically, a PEC immunosensing system is composed of a light source, reaction cell, and three-electrode-equipped working system (working, reference, and counter electrodes). Immunological compounds as biorecognition probes are first immobilized on the working photoactive electrode. After the immunoreaction process, the resulting steric hindrance effect caused by the formation of immunoconjugates with the probe increases the charge transform resistance[42]. Thus, to achieve the best performance of a PEC immunosensor, it is pivotal to build the sensing interface with efficient loading of the photoactive components and the nanobody[43].
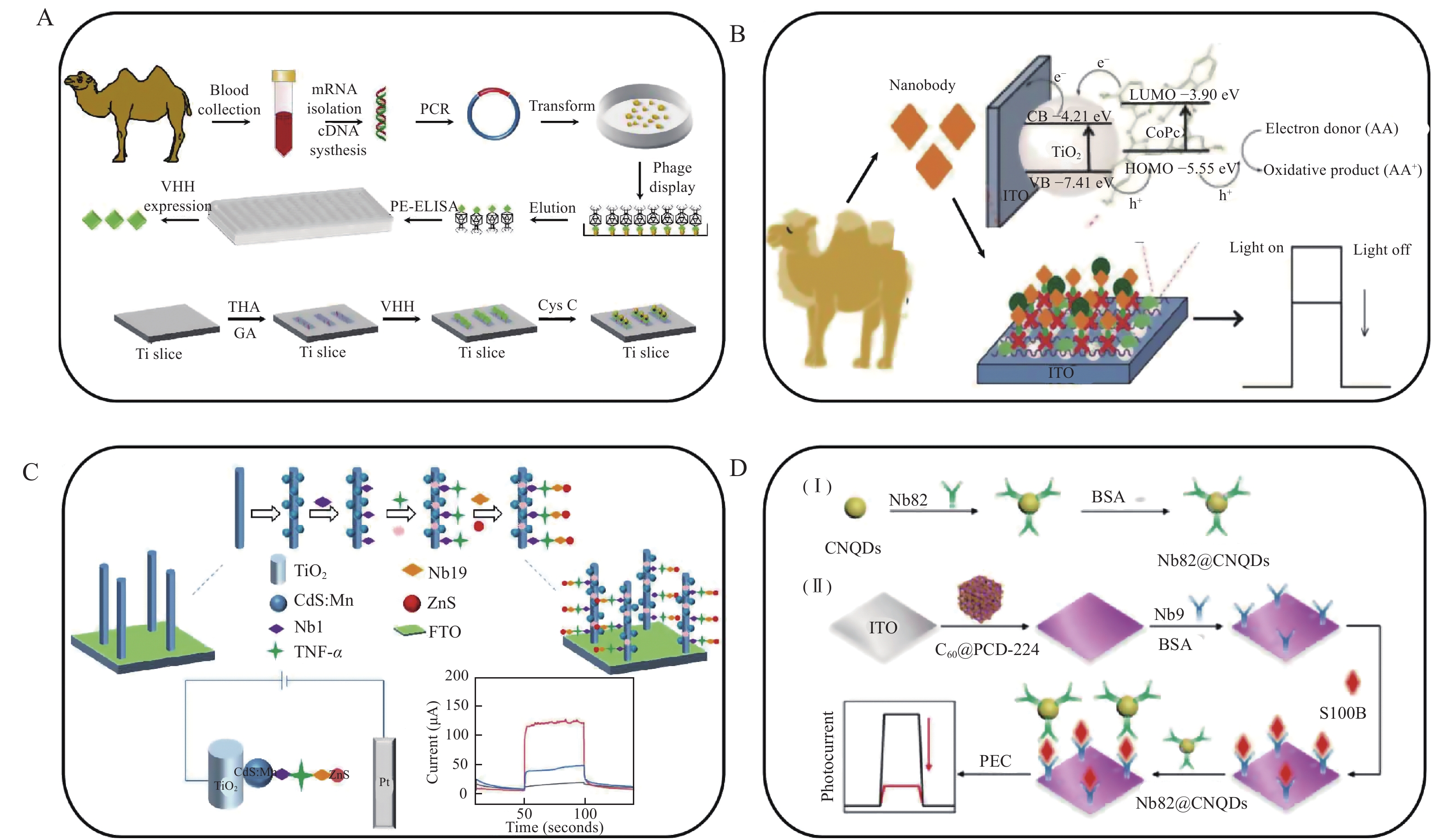
Liu et al developed a fragile PEC immunosensor combining a nanobody for the analysis of serum cystatin C, a representative biomarker for the evaluation of glomerular filtration rate and other diseases (Fig. 4A)[44]. The photoactive anode was functionalized with TiO2 nanotube arrays, providing enhanced and stable photocurrent responses owing to the uniform interfacial structure and high surface area of the TiO2 oriented on the sensing interface. After further conjugation of a detecting nanobody on the electrode, the constructed PEC immunosensor exhibited remarkable inter- and intra-assay accuracy, selectivity, and stability. A wide linear detection range was found from 0.72 pmol/L to 7.19 nmol/L with an LOD of 0.14 pmol/L.
Although TiO2 is an excellent photoactive material, its wide band gap (approximately 3.2 eV) may limit its use in a PEC module. Therefore, TiO2-based complexes or co-sensitized structures are extensively investigated to increase their photoelectric conversion efficiency[45]. Liu et al constructed a label-free PEC immunosensor using nanobody-targeting neutrophil gelatinase-associated lipocalin (NGAL), an early biomarker for acute renal failure disease (Fig. 4B)[46]. An indium−tin oxide (ITO) electrode was modified with cobalt 2,9,16,23-tetraaminophthalocyanine-sensitized TiO2, achieving five times higher photocurrent than a ITO/TiO2 electrode, and a biotinylated nanobody was immobilized on the electrode via biotin/streptavidin linkages. A low LOD of 0.6 pg/mL and a wide detection range from 0.01 to 100 ng/mL NGAL were obtained. Li et al constructed a PEC sandwich immunosensor integrating a detecting nanobody, TiO2 nanorods, and ZnS nanoparticles for ultrasensitive quantitative determination of tumor necrosis factor-α (TNF-α) in serum samples (Fig. 4C)[47]. In the assay, a TiO2 nanorod arrays/CdS:Mn2+ co-sensitized fluorine-doped tin oxide electrode was prepared. Nb1 was immobilized on the electrode surface to serve as a capture antibody, and the detecting nanobody of Nb19 was conjugated with ZnS nanoparticles to amplify the detection signals. The proposed PEC immunosensor could quantify TNF-α with a desirable calibration range from 2.0 pg/mL to 200 ng/mL with an LOD of 1.0 pg/mL.
Buckminsterfullerene (C60) has generated significant interest owing to its excellent light absorption, delocalized conjugated structure, and electron-accepting ability. However, the pristine form of C60 has poor delocalization and electron accumulation, which may limit its use in PEC immunosensors[48]. To build a biological-friendly sensing interface with extraordinary PEC response, metal–organic frameworks (MOFs) having well-regulated crystalline structures with high porosities and large specific surface areas were conjugated with C60[49]. Zhang et al reported C60 coupling with an electronically complementary porphyrin-derived MOF (i.e., PCN-224)-functionalized electrode modified with a nanobody for the determination of S100 calcium-binding protein B, a biomarker of central nervous system injury (Fig. 4D)[50]. In the assay, the detecting nanobody of Nb82 was conjugated with carbon nitride quantum dots (a dye used for bioimaging) for signal amplification. Another Nb9 was modified on the electrode as the capture antibody. It was found that the recorded photocurrent response of the immunosensor agreed well with the concentration of the antigen in the linear range of 1 to 100 ng/mL with an LOD of 0.41 pg/mL. Therefore, a PEC immunosensor using a nanobody provides insights in the new design of molecular diagnosis methods as well as instrument miniaturization.
Nanobody-based immunosensing methods are emerging analytical approaches which utilize the specific immunoreactions between an antigen and an antibody in combination with chemical analysis technologies. To achieve a rapid and sensitive immunosensing process, there have been many efforts on the molecular design and fabrication of nanomaterials in conjugation with immunoreactive agents on sensing interfaces. To maximize the bioactivity of a surface-tethered immunocomplex, different conjugation strategies, such as EDC/NHS or biotin/streptavidin coupling, glutaraldehyde crosslinking, Au–S boding, and SpyTag/Spy catcher, have been employed, in which a nanobody as a molecular probe was successfully immobilized on a platform with retention of the binding capability. Compared with a few reviews on immunoassays and immunsensors, this minireview focuses on nanobody-based immunosensing methods for public health, including disease diagnosis, environmental pollutant monitoring, and pathogenic microbe detection, which demonstrate the promising potential of a nanobody as a versatile molecular probe. Ultimately, the search for detection with increased reliability and sensitivity of biologically important substances will lead to continuous development of immunosensing systems. For example, the selection and massive production of photoelectronic-reactive nanomaterials are prerequisites for the analytical performance of PEC immunosensors. Finally, rational design and modification of nanomaterials in combination of nanobodies in immunosensing systems may present a continuous direction in this field to broaden their biomedical uses.
We thank the financial support of the National Natural Science Foundation of China (No. U1703118), Natural Science Foundation of Jiangsu Higher Education Institutions of China (No. 19KJA310003), Natural Science Foundation of Jiangsu Province (No. BK20181364), and a project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions.
| [1] |
Balayan S, Chauhan N, Chandra R, et al. Recent advances in developing biosensing based platforms for neonatal sepsis[J]. Biosens Bioelectron, 2020, 169: 112552. doi: 10.1016/j.bios.2020.112552
|
| [2] |
Jiang XS, Li DY, Xu X, et al. Immunosensors for detection of pesticide residues[J]. Biosens Bioelectron, 2008, 23(11): 1577–1587. doi: 10.1016/j.bios.2008.01.035
|
| [3] |
Prattis I, Hui E, Gubeljak P, et al. Graphene for biosensing applications in point-of-care testing[J]. Trends Biotechnol, 2021, S0167-7799(21)00011-1. doi: 10.1016/j.tibtech.2021.01.005
|
| [4] |
Yao L, He L, Yang Y, et al. Nanobiochar paper based electrochemical immunosensor for fast and ultrasensitive detection of microcystin-LR[J]. Sci Total Environ, 2021, 750: 141692. doi: 10.1016/j.scitotenv.2020.141692
|
| [5] |
Ye L, Zhao G, Dou W. An electrochemical immunoassay for Escherichia coli O157: H7 using double functionalized Au@Pt/SiO2 nanocomposites and immune magnetic nanoparticles[J]. Talanta, 2018, 182: 354–362. doi: 10.1016/j.talanta.2018.01.095
|
| [6] |
Zhang S, Chen Y, Huang Y, et al. Design and application of proximity hybridization-based multiple stimuli-responsive immunosensing platform for ovarian cancer biomarker detection[J]. Biosens Bioelectron, 2020, 159: 112201. doi: 10.1016/j.bios.2020.112201
|
| [7] |
Cheng N, Song Y, Shi Q, et al. Au@Pd nanopopcorn and aptamer nanoflower assisted lateral flow strip for thermal detection of exosomes[J]. Anal Chem, 2019, 91(21): 13986–13993. doi: 10.1021/acs.analchem.9b03562
|
| [8] |
Fan Y, Shi S, Ma J, et al. A paper-based electrochemical immunosensor with reduced graphene oxide/thionine/gold nanoparticles nanocomposites modification for the detection of cancer antigen 125[J]. Biosens Bioelectron, 2019, 135: 1–7. doi: 10.1016/j.bios.2019.03.063
|
| [9] |
Xie Y, Zhang M, Bin Q, et al. Photoelectrochemical immunosensor based on CdSe@BiVO4 Co-sensitized TiO2 for carcinoembryonic antigen[J]. Biosens Bioelectron, 2020, 150: 111949. doi: 10.1016/j.bios.2019.111949
|
| [10] |
Hu M, Wang Y, Yang J, et al. Competitive electrochemical immunosensor for maduramicin detection by multiple signal amplification strategy via hemin@Fe-MIL-88NH2/AuPt[J]. Biosens Bioelectron, 2019, 142: 111554. doi: 10.1016/j.bios.2019.111554
|
| [11] |
Zhao W, Xu Y, Kang T, et al. Sandwich magnetically imprinted immunosensor for electrochemiluminescence ultrasensing diethylstilbestrol based on enhanced luminescence of Ru@SiO2 by CdTe@ZnS quantum dots[J]. Biosens Bioelectron, 2020, 155: 112102. doi: 10.1016/j.bios.2020.112102
|
| [12] |
Ruan X, Wang Y, Kwon E, et al. Nanomaterial-enhanced 3D-printed sensor platform for simultaneous detection of atrazine and acetochlor[J]. Biosens Bioelectron, 2021, 184: 113238. doi: 10.1016/j.bios.2021.113238
|
| [13] |
Kaushik S, Tiwari UK, Pal SS, et al. Rapid detection of Escherichia coli using fiber optic surface plasmon resonance immunosensor based on biofunctionalized Molybdenum disulfide (MoS2) nanosheets[J]. Biosens Bioelectron, 2019, 126: 501–509. doi: 10.1016/j.bios.2018.11.006
|
| [14] |
Farka Z, Juřík T, Kovář D, et al. Nanoparticle-based immunochemical biosensors and assays: recent advances and challenges[J]. Chem Rev, 2017, 117(15): 9973–10042. doi: 10.1021/acs.chemrev.7b00037
|
| [15] |
Kylilis N, Riangrungroj P, Lai HE, et al. Whole-cell biosensor with tunable limit of detection enables low-cost agglutination assays for medical diagnostic applications[J]. ACS Sens, 2019, 4(2): 370–378. doi: 10.1021/acssensors.8b01163
|
| [16] |
Chen K, Xue J, Zhou Q, et al. Coupling metal-organic framework nanosphere and nanobody for boosted photoelectrochemical immunoassay of Human Epididymis Protein 4[J]. Anal Chim Acta, 2020, 1107: 145–154. doi: 10.1016/j.aca.2020.02.011
|
| [17] |
Hamers-Casterman C, Atarhouch T, Muyldermans S, et al. Naturally occurring antibodies devoid of light chains[J]. Nature, 1993, 363(6428): 446–448. doi: 10.1038/363446a0
|
| [18] |
Ingram JR, Schmidt FI, Ploegh HL. Exploiting nanobodies' singular traits[J]. Annu Rev Immunol, 2018, 36: 695–715. doi: 10.1146/annurev-immunol-042617-053327
|
| [19] |
Li SF, Zhang W, Jiang P, et al. Nanobody against the E7 oncoprotein of human papillomavirus 16[J]. Mol Immunol, 2019, 109: 12–19. doi: 10.1016/j.molimm.2019.02.022
|
| [20] |
Cyster JG, Allen CDC. B cell responses: cell interaction dynamics and decisions[J]. Cell, 2019, 177(3): 524–540. doi: 10.1016/j.cell.2019.03.016
|
| [21] |
Liu M, Li L, Jin D, et al. Nanobody-A versatile tool for cancer diagnosis and therapeutics[J]. Wiley Interdiscip Rev Nanomed Nanobiotechnol, 2021, 13(4): e1697. doi: 10.1002/wnan.1697
|
| [22] |
Anderson GP, Liu JL, Hale ML, et al. Development of antiricin single domain antibodies toward detection and therapeutic reagents[J]. Anal Chem, 2008, 80(24): 9604–9611. doi: 10.1021/ac8019398
|
| [23] |
Buser DP, Schleicher KD, Prescianotto-Baschong C, et al. A versatile nanobody-based toolkit to analyze retrograde transport from the cell surface[J]. Proc Natl Acad Sci USA, 2018, 115(27): E6227–E6236. doi: 10.1073/pnas.1801865115
|
| [24] |
De Meyer T, Muyldermans S, Depicker A. Nanobody-based products as research and diagnostic tools[J]. Trends Biotechnol, 2014, 32(5): 263–270. doi: 10.1016/j.tibtech.2014.03.001
|
| [25] |
Jaria G, Calisto V, Otero M, et al. Monitoring pharmaceuticals in the aquatic environment using enzyme-linked immunosorbent assay (ELISA)-a practical overview[J]. Anal Bioanal Chem, 2020, 412(17): 3983–4008. doi: 10.1007/s00216-020-02509-8
|
| [26] |
Li D, Morisseau C, McReynolds CB, et al. Development of improved double-nanobody sandwich ELISAs for human soluble epoxide hydrolase detection in peripheral blood mononuclear cells of diabetic patients and the prefrontal cortex of multiple sclerosis patients[J]. Anal Chem, 2020, 92(10): 7334–7342. doi: 10.1021/acs.analchem.0c01115
|
| [27] |
Schroeder HW Jr, Cavacini L. Structure and function of immunoglobulins[J]. J Allergy Clin Immunol, 2010, 125(S2): S41–S52. doi: 10.1016/j.jaci.2009.09.046
|
| [28] |
He J, Ma S, Wu S, et al. Construction of immunomagnetic particles with high stability in stringent conditions by site-directed immobilization of multivalent nanobodies onto bacterial magnetic particles for the environmental detection of tetrabromobisphenol-A[J]. Anal Chem, 2020, 92(1): 1114–1121. doi: 10.1021/acs.analchem.9b04177
|
| [29] |
Wang F, Li Z, Yang Y, et al. Chemiluminescent enzyme immunoassay and bioluminescent enzyme immunoassay for tenuazonic acid mycotoxin by exploitation of nanobody and nanobody-nanoluciferase fusion[J]. Anal Chem, 2020, 92(17): 11935–11942. doi: 10.1021/acs.analchem.0c02338
|
| [30] |
Burman B, Pesci G, Zamarin D. Newcastle disease virus at the forefront of cancer immunotherapy[J]. Cancers (Basel), 2020, 12(12): 3552. doi: 10.3390/cancers12123552
|
| [31] |
Sheng Y, Wang K, Lu Q, et al. Nanobody-horseradish peroxidase fusion protein as an ultrasensitive probe to detect antibodies against Newcastle disease virus in the immunoassay[J]. J Nanobiotechnology, 2019, 17(1): 35. doi: 10.1186/s12951-019-0468-0
|
| [32] |
Cesewski E, Johnson BN. Electrochemical biosensors for pathogen detection[J]. Biosens Bioelectron, 2020, 159: 112214. doi: 10.1016/j.bios.2020.112214
|
| [33] |
Li GH, Zhu M, Ma L, et al. Generation of small single domain nanobody binders for sensitive detection of testosterone by electrochemical impedance spectroscopy[J]. ACS Appl Mater Interfaces, 2016, 8(22): 13830–13839. doi: 10.1021/acsami.6b04658
|
| [34] |
Oloketuyi S, Mazzega E, Zavašnik J, et al. Electrochemical immunosensor functionalized with nanobodies for the detection of the toxic microalgae Alexandrium minutum using glassy carbon electrode modified with gold nanoparticles[J]. Biosens Bioelectron, 2020, 154: 112052. doi: 10.1016/j.bios.2020.112052
|
| [35] |
Zakeri B, Fierer JO, Celik E, et al. Peptide tag forming a rapid covalent bond to a protein, through engineering a bacterial adhesin[J]. Proc Natl Acad Sci USA, 2012, 109(12): E690–E697. doi: 10.1073/pnas.1115485109
|
| [36] |
Zhang M, Li G, Zhou Q, et al. Boosted electrochemical immunosensing of genetically modified crop markers using nanobody and mesoporous carbon[J]. ACS Sens, 2018, 3(3): 684–691. doi: 10.1021/acssensors.8b00011
|
| [37] |
Kissler SM, Tedijanto C, Goldstein E, et al. Projecting the transmission dynamics of SARS-CoV-2 through the postpandemic period[J]. Science, 2020, 368(6493): 860–868. doi: 10.1126/science.abb5793
|
| [38] |
Li D, Li Q. SARS-CoV-2: vaccines in the pandemic era[J]. Mil Med Res, 2021, 8(1): 1. doi: 10.1186/s40779-020-00296-y
|
| [39] |
Guo K, Wustoni S, Koklu A, et al. Rapid single-molecule detection of COVID-19 and MERS antigens via nanobody-functionalized organic electrochemical transistors[J]. Nat Biomed Eng, 2021, 5(7): 666–677. doi: 10.1038/s41551-021-00734-9
|
| [40] |
Li F, Zhou Y, Yin H, et al. Recent advances on signal amplification strategies in photoelectrochemical sensing of microRNAs[J]. Biosens Bioelectron, 2020, 166: 112476. doi: 10.1016/j.bios.2020.112476
|
| [41] |
Zhao WW, Xu JJ, Chen HY. Photoelectrochemical enzymatic biosensors[J]. Biosens Bioelectron, 2017, 92: 294–304. doi: 10.1016/j.bios.2016.11.009
|
| [42] |
Svitkova V, Palchetti I. Functional polymers in photoelectrochemical biosensing[J]. Bioelectrochemistry, 2020, 136: 107590. doi: 10.1016/j.bioelechem.2020.107590
|
| [43] |
Ge L, Liu Q, Hao N, et al. Recent developments of photoelectrochemical biosensors for food analysis[J]. J Mater Chem B, 2019, 7(46): 7283–7300. doi: 10.1039/C9TB01644A
|
| [44] |
Mi L, Wang P, Yan J, et al. A novel photoelectrochemical immunosensor by integration of nanobody and TiO2 nanotubes for sensitive detection of serum cystatin C[J]. Anal Chim Acta, 2016, 902: 107–114. doi: 10.1016/j.aca.2015.11.007
|
| [45] |
Ma X, Wang C, Wu F, et al. TiO2 nanomaterials in photoelectrochemical and electrochemiluminescent biosensing[J]. Top Curr Chem, 2020, 378(2): 28. doi: 10.1007/s41061-020-0291-y
|
| [46] |
Li H, Mu Y, Yan J, et al. Label-free photoelectrochemical immunosensor for neutrophil gelatinase-associated lipocalin based on the use of nanobodies[J]. Anal Chem, 2015, 87(3): 2007–2015. doi: 10.1021/ac504589d
|
| [47] |
Liu A, Shan H, Ma M, et al. An ultrasensitive photoelectrochemical immunosensor by integration of nanobody, TiO2 nanorod arrays and ZnS nanoparticles for the detection of tumor necrosis factor-α[J]. J Electroanal Chem, 2017, 803: 1–10. doi: 10.1016/j.jelechem.2017.09.008
|
| [48] |
Liu Y, Sheri M, Cole MD, et al. Transforming ionene polymers into efficient cathode interlayers with pendent fullerenes[J]. Angew Chem Int Ed, 2019, 58(17): 5677–5681. doi: 10.1002/anie.201901536
|
| [49] |
Liu X, Kozlowska M, Okkali T, et al. Photoconductivity in metal-organic framework (MOF) thin films[J]. Angew Chem Int Ed, 2019, 58(28): 9590–9595. doi: 10.1002/anie.201904475
|
| [50] |
Zhou Q, Li GH, Chen K, et al. Simultaneous unlocking optoelectronic and interfacial properties of C60 for ultrasensitive immunosensing by coupling to metal-organic framework[J]. Anal Chem, 2020, 92(1): 983–990. doi: 10.1021/acs.analchem.9b03915
|